Bioprocess Sampling Systems: Find the Right Sampling Approach for Your Needs

Sampling is a critical component of any drug manufacturing process regardless of modality, unit operation or stage of development.
Determining the right sampling strategy for any process depends on the process stage, sampling frequency, contamination risk, volumes required, equipment and budget available. Any one process may be limited to manual sampling or have evolved to include both manual and automated sampling. Understanding the benefits of the different technologies will help you understand how both manual and automated sampling can be complementary tools to help you monitor and control your process.
This article describes key elements of different sampling solutions to help you identify the most suitable sampling strategy for your process.
Manual and Automated Sampling
Closed sterile sampling provides the highest levels of safety and security for critical sampling steps throughout your manufacturing process. Sterile samples are collected manually with systems that do not compromise the process during sample collection and assure that the sample is representative of the process. Sterile samples are then tested off-line in a lab or testing facility. Off-line measurements require time for sample preparation and analysis and usually results in delays in data availability.
For bioburden, mycoplasma, and endotoxin testing, where samples are typically collected manually and transported away from the production environment for analysis, closed sterile sampling technologies such as NovaSeptum® GO systems or NovaSeptum® SURe assemblies are recommended (Figure 1).
Automated sampling is an essential tool for monitoring critical process parameters (CPPs) and critical quality attributes (CQAs) and the data collected is vital to the success of process analytical technology (PAT) implementation. An overarching goal of the PAT framework is to encourage more efficient strategies for development, manufacturing, and quality and to build quality into products rather than confirming at the end of the process. This science-driven, risk-based, proactive approach introduces the concept of Quality by Design (QbD) and is endorsed by the International Council for Harmonization (ICH).
In automated sampling, samples are collected at frequent intervals then transported automatically to primary or secondary destinations including analyzers. Samples require no manual intervention, and analysis is usually performed on-line or at-line close to the sampling source. Furthermore, some automated sampling solutions can sample from multiple sources and direct the samples to multiple analyzers in parallel. This approach significantly improves the speed and efficiency of sampling, enabling more efficient process monitoring and is particularly useful in a process development environment where large quantities of analytical data need to be analyzed. For GMP manufacturing, automated sampling systems such as the MAST® Autosampling Solution can accelerate moving towards real-time batch release through holistic PAT implementation (Figure 1).
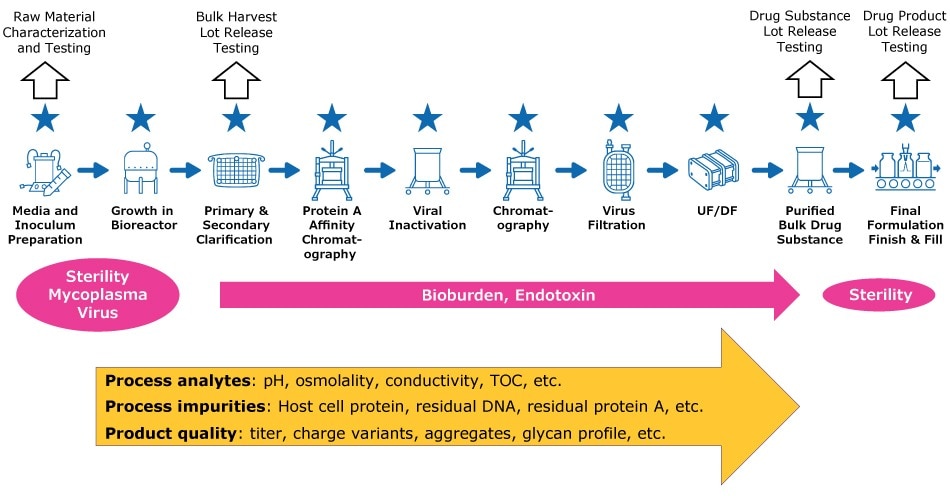
Figure 1.Schematic showing how automated and manual sampling provide complementary approaches to process sampling. Recommended sampling approaches are shown: NovaSeptum® technologies (pink), MAST® Autosampling Solution (blue stars), either technology (yellow). Dependent on release criteria, raw material and lot release testing could be performed with either technology.
For any process, selection will be guided by multiple factors including process goals, facility, infrastructure and budget available. Additional considerations that may impact selection of a sampling system are listed in Table 1.
NovaSeptum® Closed, Sterile, Manual Sampling Systems
Manual sampling using technologies such as NovaSeptum® sampling systems offer closed sampling options for both stainless-steel or single-use systems. Options for sampling stainless steel processes with the NovaSeptum® GO system include components that have been presterilized and others that are connected to tanks before cleaning in place (CIP) or steaming in place (SIP) procedures. For single-use manufacturing processes, presterilized single-use components of the NovaSeptum® SURe sampling system connect to mixers or bioreactors via C-Flex® tubing or AseptiQuick® S aseptic connectors. For each system, locking tabs and safety rings prevent accidental compromise of the system’s sterility.
Closed sterile NovaSeptum® sampling systems eliminate the risk of sample contamination and can be used throughout a GMP manufacturing process but are particularly suited to monitoring bioburden, virus, endotoxin or other purity attributes.
MAST® Automated, Aseptic, Sampling Solution
Automated sampling with technologies such as the MAST® Autosampling Solution enable on-line and near real-time measurements and can be used in both more traditional stainless steel and single-use processes. Automated sampling compresses the turnaround time between experimentation and data analysis and enables rapid access to CPP and CQA data. Faster access to critical process data is enabled through streamlined analytical integrations and makes automated sampling especially well-suited as a component of PAT. In addition, the risk either sample or source contamination due to manual handling is minimized, as are inconsistencies in test results resulting from off-line sample hold times and testing.
Currently available autosampling technologies make them ideal for real-time collection of granular process data such as metabolites, pH, conductivity, media composition, titer, among others, for both process development and manufacturing environments.
The MAST® Autosampling Solution maintains source sterility with a unique design and robust system interlocks to push samples under positive pressure, to their analytical destinations. Samples are transported through tubing that is cleaned, sanitized, and dried using high pressure after every collection cycle–all of which help mitigate contamination risk.
Comparison of Sampling Solutions
From media preparation to final filling, NovaSeptum® manual sampling solutions and MAST® Autosampling Solution provide reliable options for consistent and representative sampling and can help you demonstrate process control and assure product quality. Table 2 provides more information to help you determine the right sampling approach for your needs.
Both manual and automated sampling technologies can be used in a single process, complementing each other for improved process insights and control.
- Closed, sterile manual sampling technologies offer an easy way to confidently sample throughout your process and enable accurate sampling from a single access point.
- Automated, aseptic sampling technologies collect a sample from its source without manual intervention, enabling frequent sampling throughout the process. The sample is then directly transported to at-line or on-line analytical instruments for rapid near real-time analysis.