Separation of 2-Aminobenzamide Labelled Glycosaminoglycan Disaccharides using Zwitterionic-HILIC Column
Abstract
Glycosaminoglycans (GAGs) are polysaccharide chains that are often sulphated and are typically attached to proteoglycans. Characterization of their specific disaccharide units and sulphation patterns is crucial for understanding their functional roles and binding interactions. In this work, a method for profiling depolymerized GAG chains based on their disaccharide composition and sulphation state was developed. Separation was performed using a SeQuant® ZIC®-HILIC column. The method enabled the separation of a panel of standard disaccharides, enzymatically digested GAG polymers, and proteoglycan standards containing different sulphated disaccharide epitopes.
Section Overview
Introduction
Glycosaminoglycans (GAGs) are long chain repeating disaccharides that are generally found attached to proteoglycans. Sulphation can occur at various positions on each disaccharide pair, and these positional differences in sulphation confer distinct functional differences to their binding partners. For analytical purposes, GAGs are commonly depolymerized enzymatically into their disaccharide units (Figure 1), which are subsequently resolved chromatographically. In this study, it is demonstrated that 2-aminobenzamide (2-AB) labelled GAG disaccharide units can be resolved within a single chromatographic run using a SeQuant® ZIC®-HILIC column.1,2 In addition, GAG disaccharide profiles were obtained from proteoglycan products.
![Panel A showing glycosaminoglycan disaccharides Panel A showing glycosaminoglycan disaccharides [ΔHexAα1-3GalNAc] with sulfation variants 0S, 6S, 4S, 2S, 2S6S, 2S4S, 4S6S, 2S4S6S and hyaluronan disaccharide (HA); SNFG legend indicates unsaturated hexuronic acid (un) and N-acetylgalactosamine.](/content/dam/cms-commons/sigmaaldrich/marketing/global/images/technical-documents/articles/pharma-and-biopharma-manufacturing/small-molecules-analysis-quality-control/figure-1-a-glycosaminoglycan-disaccharide-structures.jpg)
![Panel B showing glycosaminoglycan disaccharides [ΔHexAα1-4GlcNAc] with variants 0S, 6S, NS, 2S, NS6S, 2SNS, 2S6S, 2SNS6S and hyaluronan disaccharide (HA); SNFG legend indicates unsaturated hexuronic acid (un), N-acetylglucosamine, and glucosamine. Panel B showing glycosaminoglycan disaccharides [ΔHexAα1-4GlcNAc] with variants 0S, 6S, NS, 2S, NS6S, 2SNS, 2S6S, 2SNS6S and hyaluronan disaccharide (HA); SNFG legend indicates unsaturated hexuronic acid (un), N-acetylglucosamine, and glucosamine.](/content/dam/cms-commons/sigmaaldrich/marketing/global/images/technical-documents/articles/pharma-and-biopharma-manufacturing/small-molecules-analysis-quality-control/figure-1-b-glycosaminoglycan-disaccharide-structures.jpg)
Figure 1.Glycosaminoglycan disaccharide structures after depolymerisation, including shorthand notation and Symbol Nomenclature for Glycans (SNFG).
A. Chondroitin/dermatan sulphate disaccharides (CS); B. Heparan sulphate disaccharides (HS); C. Hyaluronan disaccharides.
Experimental
Proteoglycans or GAG polysaccharides (1-2 µg each) were individually digested using an enzyme mix containing 5 mU Chondroitinase ABC (C3667) and 50 ng Heparinase I/II/III each (R&D Systems) in 100 mM ammonium acetate buffer (pH 7) in a total reaction volume of 20 µL. Digestion was carried out overnight at 30 °C. Following digestion, samples were dried in a centrifugal evaporator and labelled with 2-aminobenzamide according to an established 2-AB labelling protocol (Ludger Ltd., UK). Labelling was performed using 2-picoline borane as a reducing agent (LT-KAB-VP24), at 65 °C for 1 h. Disaccharide standards containing eight unsaturated chondroitin sulphate and eight heparan sulphate disaccharides (Iduron Ltd, UK) were also labelled with 2-AB and used as retention time standards.
Excess 2-AB was removed by octanal phase partitioning.3 Subsequently, 80 μL of the lower aqueous layer was collected and dried. The samples were then resuspended in 32 μL of 75% acetonitrile in 10 mM ammonium acetate (pH 6.8) prior to analysis by HPLC with fluorescence detection using a zwitterionic SeQuant® ZIC®-HILIC capillary column (Table 1).
Results and Discussion
Separation of 2-AB labelled sulphated glycosaminoglycan disaccharides
The retention order of 17 different GAG disaccharides according to their sulphation state on a larger dimension SeQuant® ZIC®-HILIC (3.5 μm, 150 x 2.1 mm I.D.) column has previously been established.1 Figure 2 presents a comparable separation of the same 17 GAG disaccharides using a smaller I.D. SeQuant® ZIC®-HILIC column (150 mm x 1.0 mm I.D). The figure also presents a comparison of the retention times of hyaluronic acid, eight different HS, and eight different CS disaccharides, as well as a blank (reagent only).
Gradient elution conditions were optimised for the 150 mm x 1.0 mm I.D. column, allowing all 17 disaccharides to be resolved in a 65 min multi-step gradient run at 35 °C. Compared with another popularly used method for resolving these 17 disaccharides using a C18 column with an APTS label4, zwitterionic HILIC enables a separation based on charge and, notably, by disaccharide type. When disaccharides carry an equal number of sulphate groups, chondroitin sulphate disaccharides (CS) elute earlier than heparan sulphate disaccharides (HS). This characteristic can aid in peak identification if retention time drift occurs or when unknown peaks are identified, as the 17-disaccharide panel includes only the most common types of GAGs.
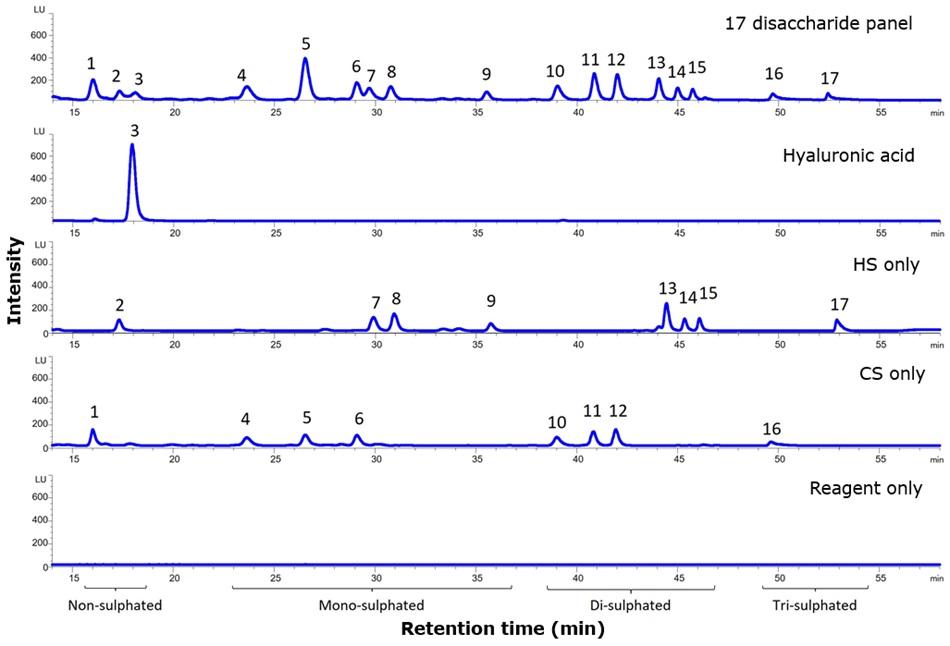
Figure 2.2-AB labelled GAG disaccharides resolved on a SeQuant® ZIC®-HILIC capillary column. CS – chondroitin sulphate, HS – heparan sulphate (peak IDs are provided in Table 3).
GAG profiles of various chondroitin sulphate and heparan sulphate products
By applying this labelling and separation technique, the sulphation profile of different proteoglycans and glycosaminoglycans can be determined. As shown in Figure 3, each GAG-containing product exhibits a distinct sulphation pattern, which may lead to differential binding of GAG-binding proteins when used in an in vitro or in vivo experiment.
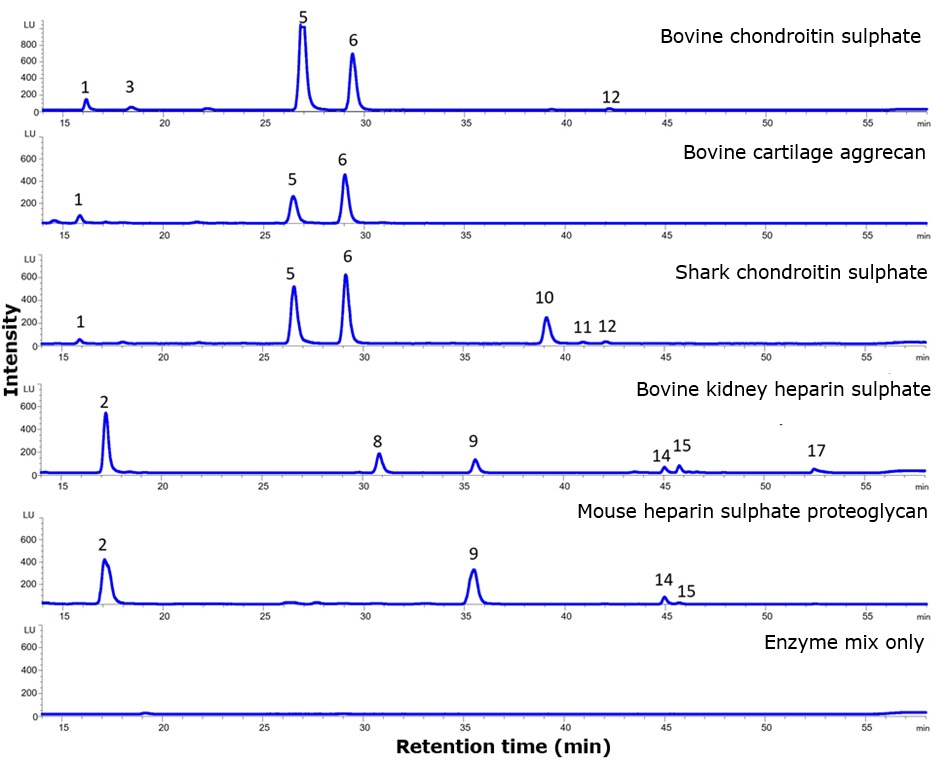
Figure 3.2-AB labelled GAG disaccharide profiles of various chondroitin and heparan sulphate products (peak IDs are provided in Table 3).