Sample Preparation by Filtration
Filtration is a separation technique used to concentrate or purify substances based on their physical or chemical properties. It is a simple and routine method used in many laboratories to remove insoluble particles from solutions and to prepare samples for analysis. Filtration is used to reduce sample complexity, improve clarity of viscous samples, and reduce background signals resulting in increased signal-to-noise ratios in analytical tests.
Featured Categories
Elevate filtration: glass, stainless steel, plastic filter holders for liquid, gas. Syringe, in-line, vacuum, pressure choices await.
Choose Millex® syringe filters: sterile, nonsterile formats. Wide membrane, housing options for HPLC, UHPLC, dissolution testing compatibility.
Seamless Sterile Filtration: Choose us for reliable Stericap® & Steripak® devices, ensuring sterile, quality solutions from mL to 20 L.
Used commonly in quantitative and qualitative analysis, filter papers must be closely matched to the application according to the physical characteristics of the filter paper. There are many sizes, materials, types, and grades of filter paper available for a variety of applications.
Depending on the filtration method applied, particles or molecules are separated based on properties such as size, shape or charge. The liquid that passes through the filter is called the ‘filtrate’ and the collected or retained material is the ‘retentate’ or ‘residue’.
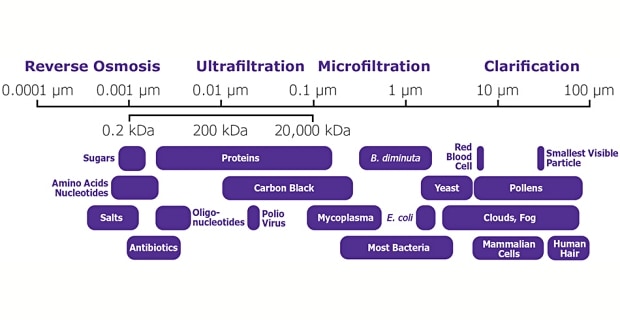
- Reverse osmosis (ionic separation) separates ions or molecules using a semipermeable membrane or barrier. Applied pressure overcomes osmotic pressure and forces solvent to move from a high solute concentration to a low solute concentration. Reverse osmosis rejects a high percentage of organic matter, other than particulates, and >99% of salts. The typical membrane rating is based on sodium chloride retention (<0.001 µm, <100 Daltons)
- Ultrafiltration (macromolecule separation) separates particles and dissolved molecules from fluids based on particle size. Ultrafiltration is used for concentration, fractionation, desalting, and buffer exchange. The typical rating is a nominal molecular weight limit (NMWL) between 1-1000 kDa.
- Microporous filtration (microfiltration) is used for particle retention/exclusion and sterilization because it separates/removes particles and biological entities, such as bacteria and cells, based on particle size. Pore sizes are typically between 0.025-10 µm and are rated as nominal (~98% retention) or absolute (100% retention of the size equal to the pore size rating).
- Clarification filters are used for pre-filtration and particle analysis because they retain/remove large particles, aggregates, and debris based on size. They may be used as a primary filtration step before microfiltration. Clarification filters typically have pore size ratings >5 µm. retained.
Common Filtration Applications
- General particulate removal
- Sample preparation for analytical techniques such as HPLC, UHPLC, ion chromatography, gas chromatography, and dissolution testing
- Sterilization of cell culture additives
- Concentration of proteins, nucleic acids and polymers
- Separation of biomolecules within a sample
- Preparation of buffers
- Water purification
Filtration is an essential sample preparation step before sensitive chromatographic analysis, such as HPLC and LC-MS. Particulates in samples can interfere with liquid, gas and ion chromatography analyses by clogging columns or column heads, or by generating contaminant peaks (“ghost peaks”) on chromatograms. Proper filtration of samples, solvents and buffers generates higher quality, more consistent analytical results. It also increases instrument uptime and prolongs column life.
Types of Filtration Processes & Procedures
There are many filters with different filtration media composition, each designed for particular applications. Filter selection depends on several factors, including:
- Size of the particles or molecules to be excluded or included
- Chemical composition of the sample
- Compatibility of the filtration media with sample or solution
- Sample viscosity
Filters can be made from different types of materials, such as paper, cloth, cotton-wool, asbestos, slag- or glass-wool, unglazed earthenware, sand, or other porous material. Membrane filters are usually made from synthetic polymers (e.g. hydrophilized PTFE, PVDF, nylon, PES).
Different forces can be applied to drive the filtration process. Filtration can be driven by simple gravity using a filter and a funnel, manually as in syringe filtration, or by centrifugal force. In vacuum-driven filtration, a vacuum pump is used to rapidly draw the fluid through a filter.
Visit our document search for data sheets, certificates and technical documentation.
Related Articles
- Syringe filter selection guide for HPLC, UHPLC, and Ion Chromatography using pore size, filter diameter, chemical compatibility, analyte binding, and extractables.
- Filtration methods and membrane filter selection in nanoparticle purification and production.
- Hydrophilic PTFE Millex® syringe filters ensure accurate dissolution testing and sample preparation for HPLC analysis.
- Ultrafiltration using Amicon® Ultra centrifugal devices for concentrating virus in preparation of high titer virus stocks and isolation of virus from samples.
- Syringe membrane filter selection and validation methods to assess analyte loss due to membrane filter adsorption in pharmaceutical quality control (QC) testing.
- See All (23)
Related Protocols
- HPLC Analysis of Polyphenols in Nero d'Avola Red Wine on Discovery® HS C18 (UV 280 nm)
- Millicup™-FLEX disposable vacuum filter was designed to reduce contamination and save time. The solvent-resistant unit allows for filtration into storage bottles.
- Samplicity® G2 filtration system enhances HPLC workflow; watch video tutorials and application notes for effective sample preparation.
- Qualitative Thin Layer Chromatography Analysis of Flavonoids and Quantification of Terpene Lactones in Ginkgo Biloba Extracts and Tablets
- HPTLC was used to analyze caffeine in coffee. Filtration of coffee through a PTFE syringe filter was the only sample preparation. Quantitation was conducted with a TLC scanner.
- See All (10)
Find More Articles and Protocols
How Can We Help
In case of any questions, please submit a customer support request
or talk to our customer service team:
Email custserv@sial.com
or call +1 (800) 244-1173
Additional Support
- Chromatogram Search
Use the Chromatogram Search to identify unknown compounds in your sample.
- Calculators & Apps
Web Toolbox - science research tools and resources for analytical chemistry, life science, chemical synthesis and materials science.
- Customer Support Request
Customer support including help with orders, products, accounts, and website technical issues.
- FAQ
Explore our Frequently Asked Questions for answers to commonly asked questions about our products and services.
To continue reading please sign in or create an account.
Don't Have An Account?



