Enrichment of Phospholipids in Biological Samples Using HybridSPE-PL
Introduction
Phospholipids (PL) are one of the major building blocks of cell membranes and are important for signal transduction and many other biologic activities. Phospholipids are abundant in biological fluids, with concentrations in human plasma as high as 1 mg/mL. The profiling of phospholipids in tissue and blood is significant in both functional and pathological studies.
HybridSPE-PL has previously been used on biological samples, such as serum and plasma, to remove phospholipid interferences prior to LC-MS analysis. We found, however, that phospholipids retained on the sorbent could be easily recovered with a strong basic solution, such as ammonium hydroxide. As such, HybridSPE-PL can also be used to enrich phospholipids for analysis and profiling. The interaction between HybridSPE-PL and phospholipids is based on Lewis acid-base chemistry and has been thoroughly discussed in a previous Reporter, volume 26.3. This article will focus on the use of HybridSPE-PL for phospholipid enrichment.
Experimental
A 100 μL aliquot of rabbit plasma was mixed with 900 μL of acetonitrile with 1% formic acid in a 1.5 mL micro-centrifuge tube and vortexed for 30 seconds, followed by centrifugation at 5000 x g for 3 minutes. The resulting supernatant was transferred to a HybridSPE-PL 96-well plate, and applied to vacuum at 10 in. Hg for 4 minutes. The flow-through was collected for LC-MS analysis. The sorbent was then washed with 1 mL of acetonitrile with 1% formic acid, and 1 mL of acetonitrile. The phospholipids retained on the HybridSPE-PL sorbent were eluted with two consecutive 1 mL aliquots of acetonitrile with 5% ammonium hydroxide. The effluent was dried down with nitrogen and reconstituted in 50% acetonitrile with 0.1% formic acid.
The sample was also prepared with a similar protocol using methanol as the main reagent. A 100 μL aliquot of plasma was mixed with 900 μL of methanol with 1% formic acid. The sorbent was washed with 1 mL of methanol with 1% ammonium formate, followed by 1 mL of methanol. The phospholipids were eluted with two 1 mL aliquots of methanol with 5% ammonium hydroxide (Figure 1 for flow chart).
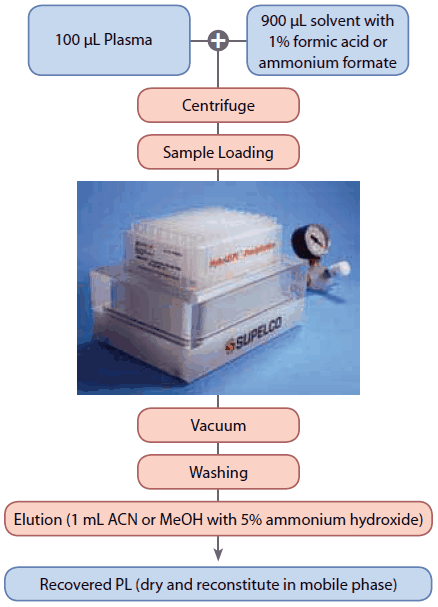
Figure 1.Experimental Flow Chart of Recovery of Phospholipids from Rabbit Plasma
LC-MS was carried out on an Applied Biosystems QTrap 3200 mass spectrometer coupled with an Agilent 1100 HPLC. A 3 μL sample was injected on an Ascentis® Express RP-Amide column (5 cm x 2.1 mm x 2.7 μm, 53911-U). The HPLC was operated in linear gradient with mobile phases A (water with 0.1% formic acid) and B (methanol: acetonitrile, 1:1/0.1% formic acid) from 50%-100% B in 10 min. The phospholipids were monitored at MRM184 and 104 after in-source fragmentation. The mass spectrometric parameters were set as follows: CUR (35), IS (5000), TEM (350), GS1 (30), GS2 (30), ihe (ON), CAD (10), and CXP (4).
Results and Discussion
The Lewis acid-base interaction between HybridSPE-PL and phospholipids can only be disrupted with a strong basic solution, such as ammonium hydroxide. Since there are high amounts of proteins in biological samples, the proteins are crashed out with organic solvents, e.g. acetonitrile with 1% formic acid or methanol with ammonium formate before enrichment. This step can be done off-line if using a cartridge, or directly in the 96-well plate. Eluted phospholipids are monitored by the mass spectrometer at product ion 184 and 104, generated by in-source fragmentation as described by Little et al.1 We observed good separation of phospholipids on a polar-embedded RP-Amide column. It should be noted that Little’s method only monitors phosphatidycholines and lyso-phosphatidycholines. We believe the same enrichment method would be applicable to other phospholipids in biological fluids since all phospholipids share the phosphate group, which interacts with the HybridSPE-PL sorbent.
Figures 2 and 3 present the LC-MS profiles of phospholipids in rabbit plasma before and after enrichment. There are two major groups of peaks on the chromatograms. The early eluted group, with retention time from 8.0 minutes to 9.0 min, is comprised of mostly single-chained phospholipids (lyso-phosphatidycholines); meaning there is only one fatty acid ester in the molecule. The later group with retention times greater than 12 minutes is comprised of mostly double-chained phospholipids; meaning there are two fatty acid esters in the molecule.
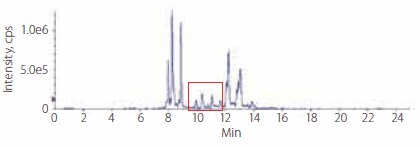
Figure 2.Profile of Phospholipids in Rabbit Plasma Before Enrichment
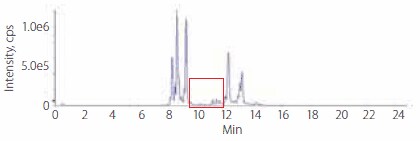
Figure 3.Profile of Phospholipids in Rabbit Plasma After Enrichment
The recovery of phospholipids is over 95% using either acetonitrile or methanol as an elution solvent. We use 1% formic acid or ammonium formate to assist in the precipitation of proteins in the plasma sample. The wash step was used to clean any soluble proteins and endogenous substances in the sample. In either method, the total loss of phospholipids is less than 5% (Table 1).
Conclusions
A simple method has been developed to enrich phospholipids from plasma samples. The method involves a HybridSPE-PL 96-well plate that both retains phospholipids and removes precipitated proteins. The interaction between the HybridSPE-PL sorbent and phospholipids is based on Lewis acid-base chemistry and can be disrupted with a strong Lewis base, such as ammonium hydroxide. The recovery of phospholipids using this method is greater than 95%. The phospholipids can be profiled on a polar embedded reversed-phase HPLC column and a triple Q mass spectrometer.
Materials
Reference
To continue reading please sign in or create an account.
Don't Have An Account?