Processing Small Tissue or Cell Samples using Sample Grinding Kit
Sample Grinding Kit is designed to disrupt cell or tissue samples. It utilizes an abrasive grinding resin and grinding pestle to rupture cells for protein extraction. Intracellular organelles are also disrupted, resulting in the liberation and extraction of all proteins soluble in the extraction solution. Samples of 100 mG or less can be processed in as little as 10 min.
The kit contains fifty 1.5-mL microcentrifuge tubes, each containing a small quantity of abrasive grinding resin suspended in water. The tube is centrifuged to pellet the resin and the water is removed. The methodology is outlined in Figure 1.
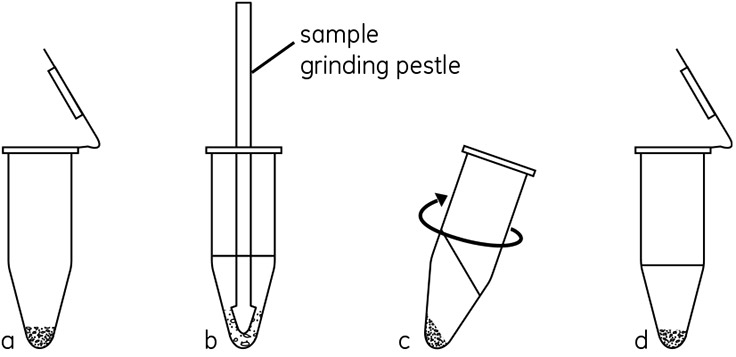
Figure 1.Schematic of the method used in the Sample Grinding Kit. (a) Pellet grinding resin in microcentrifuge tube. (b) Add sample and extraction solution. Disrupt sample by grinding with pestle. (c) Centrifuge to separate cellular debris and resin. (d) Collect supernatant.
The extraction solution of choice is added to the tube along with the sample to be ground. A disposable pestle is supplied to grind the sample. Immediately after grinding, cellular debris and grinding resin are removed by 5–10 min of centrifugation. If desired following extraction, the sample solution may be treated to remove interfering substances using 2-D Clean-Up Kit.
Protocol: Sample Grinding Kit
Components supplied
Microcentrifuge grinding tubes containing grinding resin suspended in water, disposable pestles for sample grinding.
Required but not provided
Microcentrifuge capable of at least 12000 x g, vortex mixer, extraction solution.
Preliminary notes
Samples can be extracted into 8 M urea and 4% CHAPS, or into 7 M urea, 2 M thiourea, and 4% CHAPS (see solutions A and B). Alternative nonionic detergents or protease inhibitors can be added during extraction. Carrier ampholytes (Pharmalyte™ reagents, Ampholines, or IPG Buffers) can be added at concentrations up to 2% for standard protocols but should not be added during protein extraction for labeling in 2-D DIGE.
- Briefly centrifuge the grinding resin at maximum speed in the 1.5-mL microcentrifuge tubes provided in the kit (Fig 8A). Remove supernatant with micropipette.
- Add sample (up to 100 mg) and extraction solution of choice (200–300 μL) (see solutions A and B). Tissue can be cut up with a scalpel or frozen with liquid nitrogen and broken with mortar and pestle to yield tissue fragments. Cell suspensions can be centrifuged with the grinding resin and resuspended in extraction solution.
- Grind sample thoroughly (up to 1 min) with the disposable pestle included in the kit (Fig 8B).
- Limit extraction solution to 200–300 μL during grinding to prevent liquid from splashing out of the tube. Additional extraction solution may be added to the tube following grinding (up to 1 mL).
- Separate resin and debris by centrifugation for 5–10 min at maximum speed (Fig 8C).
- Collect the supernatant and transfer to another tube (Fig 8D). If desired, proceed with further clean-up steps using 2-D Clean-Up Kit.
A. Sample preparation solution (with urea) for 2-D electrophoresis
[8 M urea, 4% CHAPS, 2% Pharmalyte or IPG buffer (carrier ampholytes), 40 mM DTT, 25 mL]
* If necessary, the concentration of urea can be increased to 9 or 9.8 M.
† Other neutral or zwitterionic detergents may be used at concentrations up to 2% (w/v). Examples include Triton X-100, NP-40, octylglucoside, and the alkylamidosulfobetaine detergents ASB-14 and ASB-16 (Calbiochem).
‡ Carrier ampholytes (Pharmalyte or IPG buffer) and DTT should be excluded from the sample extraction solution if the samples are to be labeled using 2-D DIGE. See Ettan DIGE User Manual for details.
§ Use IPG Buffer in the pH range corresponding to the pH range of the IEF separation to be performed, or Pharmalyte in a pH range approximating the pH range of the IEF separation to be performed.
Store in 2.5 mL aliquots at -20 °C.
Note: Protease inhibitors may be added if necessary.
B. Sample preparation solution (with urea and thiourea) for 2-D electrophoresis
[7 M urea, 2 M thiourea, 4% CHAPS, 2% Pharmalyte or IPG Buffer (carrier ampholytes), 40 mM DTT, 25 mL]
* Other neutral or zwitterionic detergents may be used at concentrations up to 2% (w/v). Examples include Triton X-100, NP-40, octyl glucoside, and the alkylamidosulfobetaine detergents ASB-14 and ASB-16 (Calbiochem).
† Carrier ampholytes (Pharmalyte or IPG buffer) should be excluded from the sample extraction solution if the samples are to be labeled using 2-D DIGE.
‡ Use IPG Buffer in the pH range corresponding to the pH range of the IEF separation to be performed, or Pharmalyte in a pH range approximating the pH range of the IEF separation to be performed.
Store in 2.5 mL aliquots at -20 °C.