Automated Sample Preparation for Total Drug Analysis using MultiScreen® Deep Well Solvinert Filter Plates
Measuring drug concentration in plasma or serum at different time points post-administration is essential for calculating the pharmacokinetics of a drug. We present a high-throughput, automation-compatible method for sample preparation in total drug analysis using MultiScreen® Deep Well Solvinert Filter Plates.
Request more information about MultiScreen® plates and custom plates.
Section Overview
- Concentration Determination in Total Drug Analysis
- Sample Preparation using MultiScreen® Deep Well Solvinert Filter Plates
- Determination of Drug Concentration by LC-MS/MS
- Comparison of Drug Recoveries from Filtrates Following Automated Protein Precipitation, Filtration, and LC-MS/MS Analysis
- MultiScreen® Deep Well Solvinert Filter Plates for Automated Sample Preparation
Concentration Determination in Total Drug Analysis
The determination of drug concentration in plasma or serum at various time points after administration is necessary to calculate the pharmacokinetics (PK) of a drug. Quantitative analysis of drug concentrations in plasma or serum samples using LC-MS/MS requires sample preparation using methods such as protein precipitation, solid phase extraction, or liquid/liquid extraction. Protein precipitation using a water-miscible organic solvent such as acetonitrile is the preferred protocol for high throughput workflows. Centrifugation has been the traditional method to separate precipitated proteins. However, separation by filtration has distinct benefits not offered by centrifugation, including automation compatibility and greater recovery. Filtration using a 96-well plate can improve data quality by completely separating precipitated proteins from the analyte. This is important because even the slightest particulate contamination can have a detrimental impact on LC-MS/MS analysis.
We developed an automation-compatible, high-throughput method for the determination of drug concentrations in serum or plasma samples using MultiScreen® Deep Well Solvinert Filter Plates for sample preparation. The MultiScreen® Deep Well Solvinert Filter Plate (Figure 1) is a single-use, automation-compatible 96-well filtration device that can be used to process ≤ 1.8 mL volumes of aqueous and organic solutions. The design of this filter plate allows for discrete and quantitative filtrate transfer to a standard 96-well deep well collection plate using either a vacuum manifold or a centrifuge equipped with a plate carrier. These filter plates permit in-plate mixing and incubation of acetonitrile and serum to completely precipitate serum proteins. Filtration through the 0.45 µm PTFE membrane yields a protein-free solution with complete sample recovery for seamless integration with LC-MS/MS detection.

Figure 1.Photograph of 96-well MultiScreen® Deep Well Solvinert Filter Plates.
Recoveries of three drugs (warfarin, testosterone, and propranolol) from bovine serum prepared using MultiScreen® Deep Well Solvinert Filter Plates were shown by LC-MS/MS to be highly reproducible over a broad concentration range using Tecan Genesis®, Hamilton MICROLAB® STAR and Beckman Multimek™ automation systems. Correlation of automated methods to manual and centrifugal methods is presented. These data illustrate that MultiScreen® Deep Well Solvinert Filter Plates provide a fast and effective alternative to centrifugation for sample preparation in total drug analysis.
Sample Preparation using MultiScreen® Deep Well Solvinert Filter Plates
- Warfarin, propranolol, and testosterone drug solutions were prepared by serial dilution in serum to 10, 5, 1, 0.5 and 0.1 µM in a deep well block.
- Manual protein precipitation method: A Biohit Proline™ multichannel pipettor was used to add 1000 µL of acetonitrile to each well of a MultiScreen® Deep Well Solvinert Filter Plate. Using the pipettor’s double aspiration program, 250 µL of the spiked serum solution was aspirated from the deep well block first, followed by 250 µL of acetonitrile from the filter plate to initiate protein precipitation in the pipette tip. The mixture was added back to the filter plate, which was then shaken vigorously for 2 minutes. Vacuum filtration was accomplished at >18″ Hg.
- Automated protein precipitation method: The previously described protein precipitation method was carried out separately on Tecan Genesis®, Hamilton MICROLAB® STAR, and Beckman Multimek™ workstations. The Tecan and Hamilton robots were capable of running the entire protocol, while the Beckman robot required shaking and vacuum filtration offline.
- Centrifugation: Manual protein precipitation was carried out in a deep well block shaken vigorously and spun in a Jouan CR 3.12 centrifuge at 2000 x g for 2 minutes.
Determination of Drug Concentration by LC-MS/MS
All samples were diluted to final concentrations of 1000, 500, 100, 50 and 10 nM prior to LC-MS/MS analysis. The five concentrations of each drug were analyzed in 6 replicate wells per plate. LC-MS/MS analyses were performed using a SCIEX API-2000 mass spectrometer coupled with an Agilent 1100 HPLC and well plate autosampler. A Phenomenex Synergi Hydro-RP (4 mm, 50x2 mm) C-18 column was used with a guard cartridge. LC and MS/MS conditions are listed in Table 1.
Comparison of Drug Recoveries from Filtrates Following Automated Protein Precipitation, Filtration, and LC-MS/MS Analysis
Three drugs were spiked into bovine serum followed by protein precipitation and filtration through MultiScreen® Deep Well Solvinert Filter Plates. The assay was carried out manually as well as using three robotic workstations. Drug concentrations in the filtrates were analyzed by LC-MS/MS and compared (Figure 2-4). The linearity of results for both automated and manual methods (R2 values ≈ 1) demonstrates that drug samples in serum can be reliably prepared by precipitation and filtration using a MultiScreen® Deep Well Solvinert Filter Plate (Table 2). The high degree of linearity indicates that filtrates generated following this procedure allow accurate LC-MS/MS analysis across a wide concentration range.
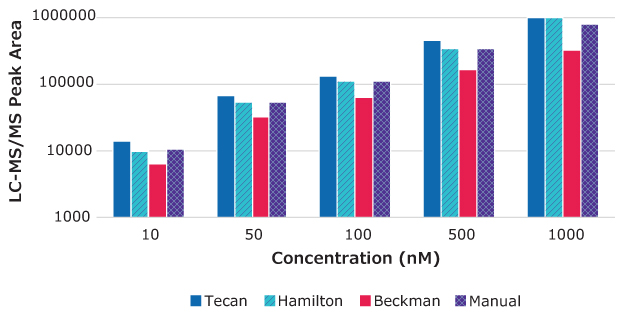
Figure 2.Peak area vs. drug concentration for warfarin samples prepared using MultiScreen® Deep Well Solvinert Filter Plates with automated and manual methods.
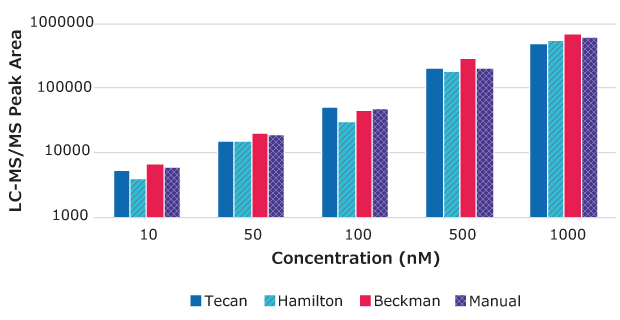
Figure 3.Peak area vs. drug concentration for propranolol samples prepared using MultiScreen® Deep Well Solvinert Filter Plates with automated and manual methods.
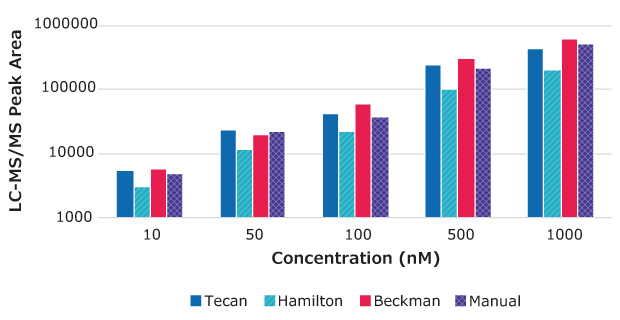
Figure 4.Peak area vs. drug concentration for testosterone samples prepared using MultiScreen® Deep Well Solvinert Filter Plates with automated and manual methods.
MultiScreen® Deep Well Solvinert Filter Plates for Automated Sample Preparation
We demonstrated an automation-compatible method for sample preparation in high-throughput total drug analysis using MultiScreen® Deep Well Solvinert Filter Plates. The MultiScreen® Deep Well Solvinert Filter Plate has several noteworthy features:
- The filter plate is fully compatible with multiple automation platforms
- Filtrates generated are free of particulates and ready for LC-MS/MS analysis
- Samples generated are highly linear across a broad concentration range
- Manual and automated methods give consistent results
- Filtration yields equivalent results to centrifugation in less time (data not shown)
- Low non-specific binding leads to high drug recovery (data not shown)
- The filter plate is compatible with most solvent systems and has no interfering extractable compounds (data not shown)