Culturing Human Aortic Smooth Muscle Cells
Read about
- Storage
- Preparation for Culturing
- Culturing HAOSMC
- Subculturing HAOSMC
- Differentiating HAOSMC
- Materials
I. Storage
Store cryovials (354-05a, 354-05f) in a liquid nitrogen storage tank* immediately upon arrival.
*Be sure to wear face protection mask and gloves when retrieving cryovials from the liquid nitrogen storage tank. The dramatic temperature change from the tank to the room could cause any trapped liquid nitrogen in the cryovials to burst and cause injury.
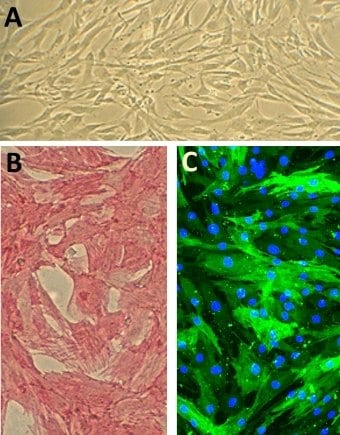
Human Aortic Smooth Muscle Cells (HAOSMC)
II. Preparation for Culturing
- Ensure the Class II biological safety cabinet, with HEPA filtered laminar airflow, is in proper working condition.
- Sterilize biological safety cabinet with 70% alcohol.
- Turn biological safety cabinet blower on for 10 minutes before beginning cell culture work.
- Ensure all serological pipettes, pipette tips and reagent solutions are sterile.
- Follow standard sterilization technique and safety rules:
- Do not pipette by mouth.
- Always wear gloves and safety glasses when working with human cells even though all strains have tested negative for HIV, Hepatitis B, and Hepatitis C.
- Complete all cell culture work in a sterile hood.
III. Culturing HAOSMC
Preparing Cell Culture Flasks for Culturing HAOSMC
- Take the Smooth Muscle Cell Growth Medium (311-500) from the refrigerator. Decontaminate the bottle with 70% alcohol in a sterile hood.
- Pipette 15 mL* of Smooth Muscle Cell Growth Medium (311-500) to a T-75 flask (SIAL0641).
*Keep the medium to surface area ratio at 1 mL per 5 cm2. For example:
- 5 mL for a T-25 flask (SIAL0639) or a 60 mm tissue culture dish (SIAL0166).
- 15 mL for a T-75 flask (SIAL0641) or a 100 mm tissue culture dish (SIAL0167).
B. Thawing and Plating HAOSMC
- Remove the cryopreserved vial of HAOSMC from the liquid nitrogen storage tank using proper protection for your eyes and hands.
- Turn the vial cap a quarter turn to release any liquid nitrogen that may be trapped in the threads, then re-tighten the cap.
- Thaw the cells quickly by placing the lower half of the vial in a 37°C water bath and watch the vial closely during the thawing process.
- Remove the vial from the water bath when only a small amount of ice is left in the vial. Do not let cells thaw completely.
- Decontaminate the vial exterior with 70% alcohol in a sterile Biological Safety Cabinet.
- Remove the vial cap carefully. Do not touch the rim of the cap or the vial with your hands to avoid contamination.
- Resuspend the cells in the vial by gently pipetting the cells 5 times with a 2 ml pipette. Be careful not to pipette too vigorously as to cause foaming.
- Pipette the cell suspension (1ml) from the vial into the T-75 flask (SIAL0641) containing 15 ml of Smooth Muscle Cell Growth Medium (311-500).
- Cap the flask and rock gently to evenly distribute the cells.
- Place the T-75 flask (SIAL0641) in a 37oC, 5% CO2 humidified incubator. Loosen the cap to allow gas exchange. For best results, do not disturb the culture for 24 hours after inoculation.
- Change to fresh Smooth Muscle Cell Growth Medium (311-500) after 24 hours or overnight to remove all traces of DMSO.
- Change Smooth Muscle Cell Growth Medium (311-500) every other day until the cells reach 60% confluency.
- Double the Smooth Muscle Cell Growth Medium (311-500) volume when the culture is >60% confluent or for weekend feedings.
- Subculture the cells when the HAOSMC culture reaches 80% confluency.
IV. Subculturing HAOSMC
Prepare Subculture Reagents
- Remove the Trypsin-EDTA solution (T3924) and Trypsin Inhibitor (T6414) from the -20 °C freezer and thaw overnight in a refrigerator.
- Make sure all the subculture reagents are thawed. Swirl each bottle gently several times to form homogeneous solutions.
- Store all the subculture reagents at 4 °C for future use.
- Aliquot Trypsin/EDTA solution (T3924) and store the unused portion at -20 °C if only a portion of the Trypsin/EDTA (T3924) is needed.
Prepare Culture Flask
- Take the Smooth Muscle Cell Growth Medium (311-500) from the refrigerator. Decontaminate the bottle with 70% alcohol in a sterile hood.
- Pipette 30 mL of Smooth Muscle Cell Growth Medium (311-500) to a T-175 flask (SIAL1080) (to be used in Section IV C Step 15.)
Subculturing HAOSMC
Trypsinize Cells at Room Temperature. Do Not Warm Any Reagents to 37 °C.
- Remove the medium from culture flasks by aspiration.
- Wash the monolayer of cells with HBSS (H6648) and remove the solution by aspiration.
- Pipette 8 mL of Trypsin/EDTA Solution (T3924) into the T-75 flask (SIAL0641). Rock the flask gently to ensure the solution covers all the cells.
- Remove 7 mL of the solution immediately.
- Re-cap the flask tightly and monitor the trypsinization progress at room temperature under an inverted microscope. It usually takes about 2 to 4 minutes for the cells to become rounded.
- Release the rounded cells from the culture surface by hitting the side of the flask against your palm until most of the cells are detached.
- Pipette 5 mL of Trypsin Inhibitor Solution (T6414) to the flask to inhibit further tryptic activity.
- Transfer the cell suspension from the flask to a 50 ml sterile conical tube.
- Rinse the flask with an additional 5 mL of Trypsin Inhibitor Solution (T6414) and transfer the solution into the same conical tube.
- Examine the T-75 flask (SIAL0641) under a microscope. If there are >20% cells left in the flask, repeat Steps 2-9.
- Centrifuge the conical tube at 220 x g for 5 minutes to pellet the cells.
- Aspirate the supernatant from the tube without disturbing the cell pellet.
- Flick the tip of the conical tube with your finger to loosen the cell pellet.
- Resuspend the cells in 5 mL of Smooth Muscle Cell Growth Medium (311-500) by gently pipetting the cells to break up the clumps.
- Count the cells with a hemocytometer or cell counter. Inoculate at 10,000 cells per cm2 for rapid growth, or at 6,000 cells per cm2 for regular subculturing.
V. Differentiating HAOSMC
Seeding HAOSMC for Differentiation
- Seed HAOSMC in the desired format at 15,000 per cm2 in Smooth Muscle Cell Growth Medium (311-500).
- Place the cells in a 37 oC, 5% CO2 humidified incubator.
- Change to Smooth Muscle Cell Differentiation Medium (311D-250) the next day by removing the growth medium from culture tissue ware by aspiration and adding the appropriate volume of Smooth Muscle Cell Differentiation Medium (311D-250). Do not allow cells to dry during medium changes.
- Incubate cells in a 37 oC, 5% CO2 humidified incubator.
Differentiating HAOSMC to Express Contractile Protein
- Remove Smooth Muscle Cell Differentiation Medium (311D-250) from culture tissue ware by aspiration. Do not allow cells to dry during medium changes.
- Add the appropriate volume of Smooth Muscle Cell Differentiation Medium (311D-250).
- Incubate cells in a 37 oC, 5% CO2 humidified incubator in the Smooth Muscle Cell Differentiation Medium (311D-250).
- Change to fresh Smooth Muscle Cell Differentiation Medium (311D-250) every other day.
- HAOSMC are in growth arrest and smooth muscle a-actin is expressed in 10 days.
Related Products
Loading
Sign In To Continue
To continue reading please sign in or create an account.
Don't Have An Account?