Membrane Chromatography
Our Natrix® chromatography membrane technology provides a new level of downstream productivity and versatility. The macroporous structure, combined with high functional group density, enables high load capacity and excellent impurity removal at ultra-fast flow rates. With great performance in a single-use and scalable format, our innovative Natrix® products improve process economics and facility utilization.
Products

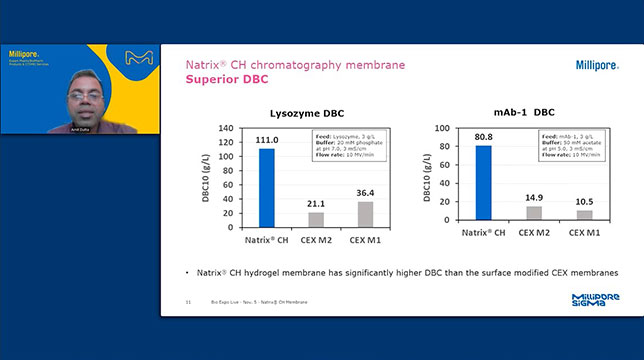
Natrix® CH chromatography membrane
Chromatographic purification is a frequent bottleneck in biomolecule purification. With high load capacity and ultra-fast flow rates, our Natrix® chromatography membranes deliver excellent impurity removal in a scalable, single-use format. This innovative technology solves many of your purification challenges and enables a new level of downstream productivity while improving process economics and facility utilization.

Natrix® Q chromatography membrane
A high-capacity, high-throughput, strong anion exchange membrane chromatography device designed for flow-through purification of biomolecules and removal of endotoxins, viruses, HCP and nucleic acids.
Natrix® Q chromatography membrane is a porous polyacrylamide hydrogel containing a high density of pendant quaternary ammonium (Q) binding groups directly polymerized within an inert, macroporous membrane scaffold. The interconnected pore structure and high functional group density enables fast flow rates with high loading capacity while maintaining excellent impurity removal across a wide range of buffer conditions. Natrix® Q chromatography membrane devices improve your productivity, flexibility, economic utility and process robustness.
Related Resources
- Data Sheet: Natrix® CH Chromatography Membrane
For single-use flow through aggregate removal.
- Application Note: Higher Volume Processing Using Natrix® Q Device
This application note describes a series of tests to assess performance of a Mobius® tubing assembly containing two Natrix® Q Process 600 devices manifolded in parallel.
- Article: Cation Exchange Chromatography Membrane for mAb Purification
The Natrix® CH chromatography membrane device achieves high binding capacity at fast flow rates for accelerated mAb purification.
- FAQ: Natrix® CH Chromatography Membrane
This document lists all the frequently asked questions about Natrix® CH devices.
- Guide: Biochromatography Portfolio Guide
Bulk Resin, Membranes and Prepacked Columns
- Guide: Chromatography Product Selection Guide
Protein A Affinity Chromatography Options
- Guide: Natrix® Q Chromatography Membrane
With a revolutionary three-dimensional macroporous hydrogel structure that provides a high density of binding sites and rapid mass transfer, Natrix® membranes deliver binding capacity that exceeds resin-based columns with fast flow rates.
- Data Sheet: Natrix® Q Chromatography Membrane - For single-use, scalable purification
Natrix® chromatography membrane is a high capacity, high throughput strong anion exchange device designed to purify biomolecules.
- Guide: Natrix® Q Chromatography Membrane Best Practices Guide
With an innovative macroporous hydrogel structure that provides a high density of binding sites and rapid mass transfer, Natrix® Q anion exchange (AEX) chromatography membranes deliver higher binding capacities than resin-based columns at fast flow rates typical of traditional membrane adsorbers.
- Application Note: Comparison of process-related impurity clearance for membrane adsorbers and resin-based chromatography technologies
Strong anion exchange (AEX) chromatography has become an industry standard in polishing steps for monoclonal antibody (MAb) purification.
- Application Note: Robust, Load Independent Viral Clearance in Monoclonal Antibody Purification with Natrix® Q Chromatography Membrane
Anion exchange chromatography is an important flowthrough polishing step for viral clearance of monoclonal antibodies (mAbs).
- Brochure: What's Next: Exploring the Future of Bioprocessing Together
Respondents viewed intensified technologies as mostly single use at the clinical-stage and as mostly hybrid at the commercial scale.
- Article: Purification of Extracellular Vesicles with Eshmuno® Q Resin and Natrix® Q Membrane
Separation and purification of subpopulations of extracellular vesicles (EVs) using Eshmuno® Q anion exchange resin and Natrix® Q membrane chromatography.