Lateral Flow Test: Design, Materials & Manufacturing Insights
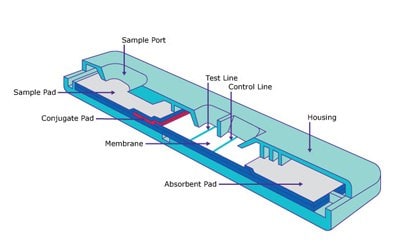
Figure 1.Lateral flow test strip design
Lateral flow test strips have revolutionized the field of diagnostics, offering quick and convenient testing for a wide range of analytes. Originally developed for detecting human chorionic gonadotropin (hCG), these test strips now cover applications in ovulation monitoring, infectious disease detection, drug analysis, and more. The COVID-19 pandemic further boosted the popularity of rapid antigen testing, leading to the emergence of at-home testing products utilizing lateral flow technology. We aim to provide more insights into the design, materials, reagents, and manufacturing processes involved in developing immunochromatographic test strips. By understanding these principles, you can streamline the test development process and ensure reliable and consistent performance.
Why is nitrocellulose membrane used for lateral flow assays?
Nitrocellulose membrane is commonly used in lateral flow assays due to its unique properties. It acts as a solid support, immobilizes various components of the assay, and allows capillary flow in a reasonable time. Here are some reasons why nitrocellulose membrane is preferred:
- Capillary action: Lateral flow nitrocellulose membranes exhibit excellent capillary action, which allows the sample to flow through the test strip without the need for external pumps or power sources.
- Binding capacity: Nitrocellulose has high protein-binding capacity, making it suitable for adsorbing analytes or antibodies. This property enables the immobilization of specific molecules at different zones on the membrane to create the test line and control line.
- Capillary flow rate: Lateral flow nitrocellulose membranes are available with different flow rates, allowing for the design of a test strip taking into consideration the required sensitivity, specificity, and time to result.
- Compatibility: Nitrocellulose is compatible with various assay reagents, buffers, and detection methods.
- Membrane wicking: The membrane's wicking properties support the movement of fluids across the detection zones, facilitating signal development and minimization of background.
Overall, the use of nitrocellulose membrane in lateral flow assays provides a reliable and cost-effective platform for rapid diagnostic testing across multiple applications.
What are the recommended storage conditions for Hi-Flow™ Plus membranes?
Hi-Flow™ Plus membranes, made of nitrocellulose, should be stored under specific conditions to maintain their functionality. It is important to protect them from exposure to organic solvent vapors as these can render the membranes hydrophobic. They should remain sealed in their foil pouches to prevent dirt and dust penetration. For long-term storage, it is recommended to store them at a temperature between 10 and 25 °C with a relative humidity ranging from 30 to 70%, avoiding condensing atmospheres and direct sunlight. Due to the highly flammable nature of nitrocellulose, precautions against fire must be implemented in the storage area. Please note that the conditions for long-term storage differ from the precise control required in manufacturing areas, and the membrane stability profile may not predict the overall stability of finished test strips.
What are the essential instruments for developing a rapid lateral flow test?
Essential instruments for developing a rapid lateral flow test include a precision dispensing system for spraying capture reagents onto the membrane and a precision cutting system for converting master cards into individual test strips. These processes cannot be completed manually with the reproducibility required for consistent test strip performance.
What are capture reagents in lateral flow test strips?
Capture reagents are essential components of lateral flow test strips that enable the detection of target molecules. Bound to the nitrocellulose surface, they are the starting point for formation of multimolecular complexes at the test and control lines, indicating the presence or absence of the target molecule. Antibodies are commonly used as capture reagents due to their high affinity and specificity. The format of many lateral flow tests is based on immunoassays, with different configurations depending on the type and size of the target molecule.
In sandwich assays, the target molecule acts as a bridge between the capture antibody bound to the membrane and a second capture antibody conjugated to a detector particle. Competitive and inhibition assays utilize a single capture antibody and the target molecule, often conjugated to a carrier molecule. In a competitive assay, free target molecules in the sample compete with target molecules conjugated to the detector particle for binding to the capture antibody.
In an inhibition assay, target molecules in the sample bind to capture antibodies conjugated to the detector particles, thereby inhibiting their ability to bind to target molecules immobilized on the membrane. In serum assays, where the target molecule is an antibody, the capture molecules can be antigens specific to the antibody being detected. Recently, nucleic acid detection has been explored, using labeled primers in a PCR reaction as recognition sites for capture reagents. The choice of the primer labels and corresponding capture reagents depends on consideration of the desired sensitivity, stability, purity, and availability.
How can screening strategies optimize the development of rapid lateral flow tests?
To optimize the development of rapid lateral flow tests, screening strategies play a critical role. Here are some key aspects to consider:
- Orthogonal Approach: An orthogonal approach should be taken to evaluate all possible combinations of antibodies or capture reagents for detecting the target molecule. This systematic evaluation helps identify the most suitable pairs with the highest potential for further optimization.
- Testing Matched Pairs: Even if antibodies are labeled as a "matched pair" by the supplier, they must be tested within the lateral flow test strip format. This testing determines which antibody is best suited for application to the membrane and which is best for conjugation to the detector particle. Matched pairs identified in an ELISA format may not necessarily work optimally in a lateral flow test strip.
- The "Half-Stick" Assay: The "half-stick" assay is a simplified approach used for initial screening. Using a 96-well plate, a strip consisting of only a membrane and absorbent pad is placed in a well containing detector particles and the target molecule in a sample buffer. The liquid is allowed to run up the half-stick until fully absorbed, and then the half-stick is transferred to a second well with sample buffer alone. This process helps minimize background by washing excess detector particles into the absorbent pad.
- Optimization Opportunities: The half-stick assay provides an opportunity to optimize various components of the lateral flow assay. These include capture antibody buffer conditions, membrane drying parameters, membrane blocking methods, detector particle properties (e.g., amount, conjugation process, antibody load, blocking agent), and sample buffer composition.
By employing these screening strategies and optimizing the assay components, developers can enhance the performance, reliability, stability, and shelf life of rapid lateral flow tests.
What are the different manufacturing strategies for lateral flow assays?
Lateral flow assay manufacturing can be done either manually, through automation, or a combination of both. Manual processes involve batch processing, while automation allows for continuous processing. The choice of strategy depends on factors such as production volume and available resources. Both manual and automated methods can yield high-quality products. Batch processing equipment is less expensive, while labor costs are lower in fully automated systems. Matching the lot sizes of materials and reagents during the planning process minimizes waste.
How does the chosen manufacturing process affect the dimensions of the materials used in lateral flow assays?
The manufacturing process chosen impacts the dimensions of the materials. Batch processing typically uses membrane strips or sheets with well-defined length and width requirements and small tolerances. Membrane rolls may also be used but usually require cutting into strips during manufacturing. For striping, the membrane should be flat and not overly rigid. Some plastic backings may be too thick and unsuitable. Reel-to-reel processes require rolls of materials. Edge alignment, edge quality, telescoping, and packaging requirements also need to be considered.

What are the manufacturing considerations regarding the physical structures of the materials used in lateral flow assays?
Manufacturing processes and equipment can potentially impact the physical structures of the materials used in lateral flow assays. Care must be taken to ensure the materials are not damaged during manufacturing or handled in ways that make their performance unpredictable. Attention to detail is vital, and in-depth knowledge of the test strip mechanisms is required to achieve consistency in manual assembly operations. Continuous training of manufacturing and quality control personnel, along with in-process inspection, helps minimize waste and maximize yield. Unbacked membrane rolls have a low tensile strength making them challenging to handle without breaking. Backed membranes are recommended for continuous processes.
What are some manufacturing strategies for optimizing the capture reagent buffer in rapid lateral flow tests?
Manufacturing strategies for optimizing the capture reagent buffer in rapid lateral flow tests include adjusting the concentration of the capture reagent, adding stabilizers to the buffer, and using a different buffer system altogether. These strategies can help improve the stability and shelf life of the test. It is important to account for the capture reagent solution is dried onto the membrane. Buffer formulations that are acceptable for entirely aqueous assays may be unsuitable for lateral flow test strips if evaporation of the water leads to a chemical environment that interferes with adsorption to the nitrocellulose or denatures the capture reagent.
How do membrane selection and specifications impact the sensitivity of a rapid lateral flow test?
Membrane selection and specifications can significantly impact the sensitivity of a rapid lateral flow test. The thickness, backings, and flow rate of the membrane can all affect the flow of the sample and the binding of the capture reagents, leading to changes in sensitivity.
There is a direct correlation between the speed and sensitivity of the membrane. Understanding these requirements will enable you to select the appropriate membrane for your application.
What is the purpose of a product definition in the development of lateral flow tests?
A product definition is essential in the development of lateral flow tests to establish the basic product qualities and ensure that resources are not spent on a design that is unfeasible or impractical.
What are the key aspects of immunochromatographic test strip design?
The key aspects of immunochromatographic test strip design include the selection of appropriate materials, the integration of reagents, and the optimization of manufacturing processes to ensure consistent performance and accuracy.
How can uncharacterized variations in a single material, reagent, or process affect the consistency of performance of a lateral flow test?
Uncharacterized variations in a single material, reagent, or process can lead to inconsistent performance of a lateral flow test, potentially resulting in false negatives or false positives. It is essential to carefully control and characterize all materials, reagents, and processes used in test strip manufacture to ensure consistent performance.
What is the importance of reproducibility in diagnostic test strip development?
Reproducibility is critical in diagnostic test strip development, as the final product must be consistent in its performance if it is to be commercially successful. A high level of reproducibility should be a design goal from the outset of product development.
Can I purchase Hi-Flow™ Plus membranes online?
Yes, Hi-Flow™ Plus membranes for manufacturing scale purposes are now available to purchase online in convenient pack formats of 20mm and 25mm wide.
See the products table below for our offering.
Looking to gain even more insights into the design, materials, reagents, and manufacturing processes involved in developing lateral flow assay test? Sign up for our Rapid Lateral Flow Tests Guide.
To continue reading please sign in or create an account.
Don't Have An Account?