Automated, High Throughput Sampling and Process Analytical Technology

The concept of process analytical technology (PAT) is to encourage the use of more efficient strategies for development, manufacturing, and quality assurance by the biopharmaceutical industry. A key objective is to build quality into products rather than evaluating quality at the end of the process. 1-4
When integrated into development and manufacturing workflows, PAT enables significant improvements in process understanding and supports the development of dynamic manufacturing processes through real-time monitoring, accelerated control through feedback loops, real-time quality assurance, and real-time release.
In this technical article, you can learn more about sampling strategies to support process monitoring.
To learn more about PAT in general, explore our technical article Improving Efficiency and Control in Biopharma Manufacturing With Process Analytical Technology.
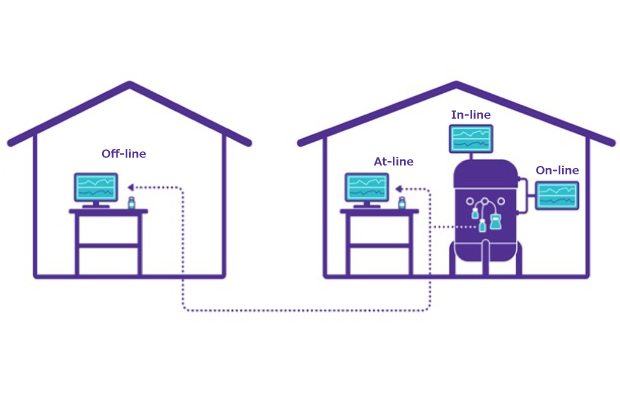
Figure 1.Types of sampling to support PAT
Sophisticated PAT for monitoring upstream cell culture processes is increasingly being adopted and includes measurements for pH, dissolved oxygen, carbon dioxide among others. In addition, advanced spectroscopic or chromatographic methods have been developed for in-line or at-line measurements of process parameters for both upstream and downstream workstreams, including sensors to measure host cell proteins, protein aggregates, and more. As manufacturers move towards process intensification and additional technologies are developed for real-time measurement of critical control parameters (CPPs), high-throughput automated sampling will become an integral component of biopharma manufacturing. Table 1 describes the types of analysis from these tests and highlights the difference in the shift from standard off-line testing to on-line, in-line, and at-line tests.
On-line and in-line tests provide more dynamic, real-time analysis and offer faster response times as compared to off-line measurements where samples are collected, analyzed off-site and usually have time delays between the point of sampling and results.
To learn more about automated aseptic sampling watch our on-demand webinar Automated Aseptic Sampling in Bioprocessing,
Data-driven Sampling and Process Improvement
Continuous process improvement includes development, process validation, and life cycle management, all of which are facilitated by sampling.
For process development, it is important to quickly analyze process intermediates and processing conditions to avoid extending the process time that could impact results. It is also important to understand critical material CPPs that impact the drug critical quality attributes (CQAs). During process validation, the design space is defined based on critical parameters. In terms of lifecycle management, sampling at key points is essential for process monitoring and identifying process trends, but these are only possible if samples are collected at the right frequency from the right points in the process with quick turnaround of analytical results.
Traditional off-line manual sampling methods can present challenges related to sample source contamination, inadequate sample traceability and delivery, lengthy analytical turnaround times, and inconsistent results. They are also resource heavy, have long durations from sampling to results, and require the utmost care from operators in data recording and disposition.
Automated sampling systems overcome these challenges by automatically collecting samples at frequent or process-based sampling points without manual intervention. Samples are transferred by the sampling system to a temporary sample preparation and storage equipment or directly to analytical destinations for analysis, accelerating access to process and quality data points. These sampling technologies can support multiple sample sources and multiple analytical destinations, enabling simultaneous analysis of a variety of parameters, further speeding up process insights. Automated sampling systems enable near real-time, on-line measurements of CPPs and CQAs at any process scale and integrate quality directly into the GMP manufacturing process.
These automated sampling systems are an essential PAT component and promote the collection of more consistent, process representative data.
Monitoring with Manual or Automated Sampling Technologies
Off-line monitoring, where samples are collected manually and moved to outside labs for testing or stored for future analysis, relies on sampling technologies such as the NovaSeptum® Sampling Solutions. These reliable, closed, sterile sampling solutions can be used throughout large-scale GMP manufacturing workflows and eliminate the risk of either process or sample contamination. Importantly, they are ideally suited for monitoring bioburden, virus, endotoxin, or other purity attributes, particularly where samples need to be collected, transported, or stored for extensive periods.
However, for more frequent on-line or at-line monitoring to retrieve real-time process data with minimal operator involvement, automated sampling may be the preferred choice. The MAST® Autosampling Solution can be integrated into process development environments where large amounts of analytical data is beneficial to process understanding and optimization, or to GMP manufacturing to improve process control and product quality.
Real-time, on-line measurements of granular process data such as metabolites, pH, conductivity, media composition, titer, among others, can be quickly collected and enable more accurate predictions of what is occurring at the sample source, improves process understanding, and speeds up decision making. Read our technical article to explore how continued process verification (CPV) helps drug manufacturers ensure their processes remain in a continual state of control so that the strength, quality, and purity of the final drug product is maintained.
For additional information on sampling solutions, please visit Bioprocess Sampling Systems: Find the Right Sampling Approach for Your Needs.
