Kinetic Performance Benefits of a Superficially Porous Particle Zwitterionic HILIC Column
Abstract
The 2.7 µm superficially porous particle (SPP) Ascentis® Express ZIC®-cHILIC column was compared with a 3 µm fully porous particle (FPP) SeQuant® ZIC®-cHILIC column. Enhanced kinetic performance was observed for the Ascentis® Express column, as indicated by reduced plate heights and lower resistance to mass transfer. This enables more efficient separations and/or faster analyses while maintaining backpressures comparable to those of the FPP, thereby supporting optimized separations and improved throughput in HILIC applications.
Section Overview
Introduction
The SeQuant® ZIC® family of HPLC products has gained widespread popularity for separations in hydrophilic interaction liquid chromatography (HILIC). These HPLC columns utilize a polyzwitterion grafted to high purity type-B silica particles, forming a thick layer of surface adsorbed water that is required for highly selective separations in HILIC mode. However, these chemistries have historically been available only on 5 µm, 3.5 µm, and 3 µm fully porous particle (FPP) sizes which have limited their chromatographic performance to traditional HPLC efficiencies. To obtain UHPLC performance, either smaller sub-2 µm particles can be employed or superficially porous/Fused-Core® particles can be used. However, the use of sub-2 µm particles typically results in higher backpressures. In contrast, superficially porous particles (SPP) provide an alternative approach to achieving UHPLC performance and form the basis of the technology used in the Ascentis® Express particles. Often also referred to as pellicular or solid-core particles, these particles are characterized by a solid, nonporous core surrounded by a porous outer shell, as illustrated in Figure 1. The performance benefits of these particles, compared with fully porous particles have been well documented.1-6 In simplified terms, these particles offer UHPLC column efficiencies similar to those of smaller fully porous particles, but with backpressures comparable to those of FPP of the same particle size.

Figure 1.a. Fully porous particle – FPP; b. superficially porous particle-SPP.
To modernize the use of zwitterionic stationary phases for UHPLC HILIC separations, a new development that combines 2.7 µm Ascentis® Express particle with the ZIC®-cHILIC phase was introduced. These new Ascentis® Express ZIC®-cHILIC particles feature the same polyphosphorylcholine (PC) grafted surface, but with a slightly wider pore size of 160 Å. This modification provides high efficiencies and improved kinetic properties that enable faster separations.
In this work, the new 2.7 µm SPP, 160 Å Ascentis® Express ZIC®-cHILIC column is compared with the standard 3 µm FPP, 100 Å SeQuant® ZIC®-cHILIC column. The performance gains of the Ascentis® Express ZIC®-cHILIC are highlighted.
Experimental
The Knox Plot approach (Equation 1) was used to compare the kinetic performance of the two columns by plotting h (reduced plate height) against v (reduced interstitial velocity). Additional information on kinetic plots in HPLC can be found in the cited literature.7-12 The key parameters and terms are summarized in Table 1 to aid in interpretation of the Knox plot.
Equation 1. Knox Equation: h = Av1/3 + B/v + Cv
The SeQuant® ZIC®-cHILIC columns are commonly packed in a PEEK-lined stainless-steel tube with titanium frits whereas the Ascentis® Express ZIC®-cHILIC is housed in a bio-inert coated stainless-steel tube and frits (INERTProve™ hardware). To alleviate potential differences in flow dynamics arising from the PEEK-lined hardware and different frit configurations at high flow rates, the SeQuant® ZIC®-cHILIC particles were packed into an identical INERTProve™ hardware, allowing a fair comparison of the packed bed characteristics between the two different particle types. Cytidine was selected as the probe compound for comparing column performance because its diffusion coefficient has been experimentally determined, which is required for comparisons under the reduced conditions of the Knox plot.13 The retention factor was adjusted to 2.0 ±0.1 for both columns by selecting appropriate mobile phase conditions (Table 2). The void volume markers, phenyldodecane and decanophenone, were carefully selected based on recommendations for more accurate void volume determination in HILIC depending on the mobile phase conditions.14
The Ascentis® Express ZIC®-HILIC showed lower reduced plate heights (Figure 2A), with a flatter increase at higher flow rates (reduced A & C terms in the Knox equation), while exhibiting similar backpressure behaviour to the SeQuant® ZIC®-cHILIC (Figure 2B). The significantly improved efficiency of the superficially porous particle-packed Ascentis® Express column is reflected in the plates per meter achieved across the flowrate range shown in Figure 2C. These results indicate expanded options for more efficient separations at higher flow rates and the potential for faster, higher resolution separations, enabling higher sample throughput.
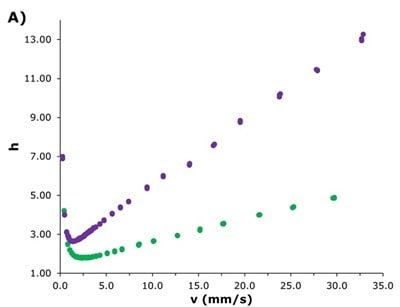
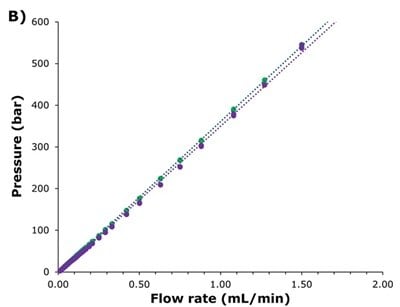
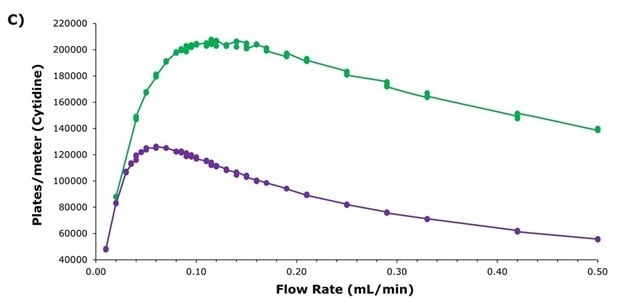
Figure 2. Comparisons of Ascentis® Express ZIC®-cHILIC (green) to SeQuant® ZIC®-cHILIC (purple). A) Reduced plated height vs. reduced velocity plot, B) Pressure profile across the tested flowrates, and C) Plates/meter comparison across conventional flow rates used for HILIC separations with a 2.1 mm I.D. column.
To illustrate the potential time gain, Figure 3 compares chromatograms obtained with the SeQuant® ZIC®-cHILIC column at its minimum reduced plate height (hmin) with those obtained using the Ascentis® Express ZIC®-cHILIC column at the same reduced plate height (hmin = 2.67), as well as at its own minimum reduced plate height (hmin = 1.79). At this equivalent reduced plate height (hmin = 2.67), the Ascentis® Express ZIC®-cHILIC column operated at a significantly higher flow rate, providing a 7.5x time saving while still achieving a higher plates per meter value for cytidine (138,537 vs. 126,327). This result highlights the potential of Ascentis® Express ZIC®-cHILIC column to significantly accelerate UHPLC HILIC applications.
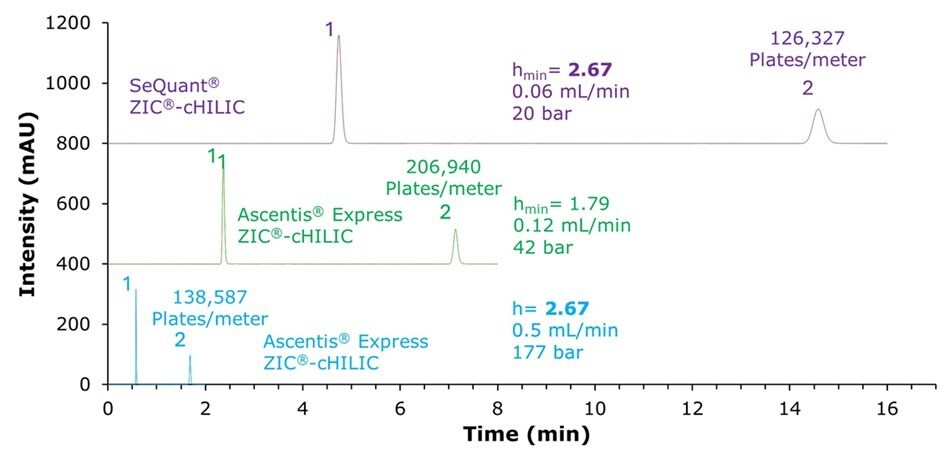
Figure 3.Comparison of chromatograms obtained with the SeQuant® ZIC®-cHILIC column (purple) at its minimum reduced plate height (hmin = 2.67) at a flow rate of 0.06 mL/min (20 bar) and with the Ascentis® Express ZIC®-cHILIC column (green) at its own minimum reduced plate height (hmin = 1.79) at 0.12 mL/min (42 bar) and at the same reduced plate height (turquoise) as the SeQuant® ZIC®-cHILIC column (hmin = 2.67) at 0.50 mL/min (177 bar). Peaks: (1) decanophenone for SeQuant® ZIC®-cHILIC or 1-phenyldodecane for Ascentis® Express ZIC®-cHILIC, (2) Cytidine
Conclusion
The new 2.7 µm superficially porous particle, 160 Å Ascentis® Express ZIC®-cHILIC column demonstrated superior column performance compared with the classic 3 µm fully porous particle SeQuant® ZIC®-cHILIC column. This improvement arises from a significant reduction in the A-term (eddy diffusion or multi-path term), which results in a much lower overall reduced plate height at nearly equivalent pressure drops. Additional improvements were observed in the C-term (resistance to mass transfer) of the Knox function, which was approximately threefold smaller than that of the fully porous particles, enabling higher flow rates with less loss in column efficiency. This performance enhancement makes the Ascentis® Express ZIC®-cHILIC column well suited for chromatographers seeking faster UHPLC HILIC separations, where some efficiency may be traded for considerable throughput gains at system pressures compatible with standard HPLC equipment.
Related Products
HPLC Columns
Solvents, Reagents, and Reference Materials
References
To continue reading please sign in or create an account.
Don't Have An Account?