UHPLC Analysis of Methotrexate and Related Compounds on Ascentis® Express C18
CONDITIONS
column
Ascentis Express C18, 10 cm x 2.1 mm I.D., 2.0 μm particles (50813-U)
mobile phase
[A] 10 mM ammonium formate, pH 3.0 with formic acid; [B] acetonitrile; (90:10, A:B)
flow rate
0.7 mL/min
pressure
1153 psi (795 bar)
column temp.
35 °C
detector
UV, 254 nm
injection
1 μL
sample
50 μg/mL in 85:15, water:methanol
Description
Analysis Note
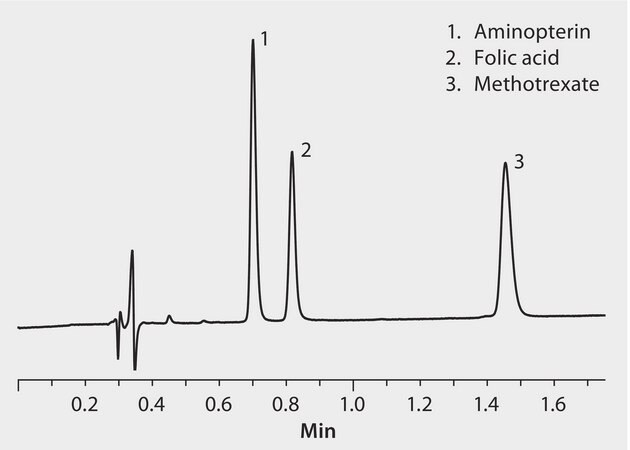
Methotrexate (MTX) has been selected as a tracer of surface contamination exposure of hospital staff to antineoplastic drugs (ANDs). Its analysis is an emerging facet of occupational medicine in hospital premises. Methotrexate and the related compounds aminopterin and folic acid are shown here separated by UHPLC on an Ascentis Express C18 column in less than two minutes. The rapid, high resolution separation means the method can be employed in hospitals or clinical settings to routinely monitor the contamination levels of surfaces.
Legal Information
Ascentis is a registered trademark of Merck KGaA, Darmstadt, Germany