3D Organoid Culture: New In Vitro Models of Development and Disease
2D vs. 3D Cell Model Systems
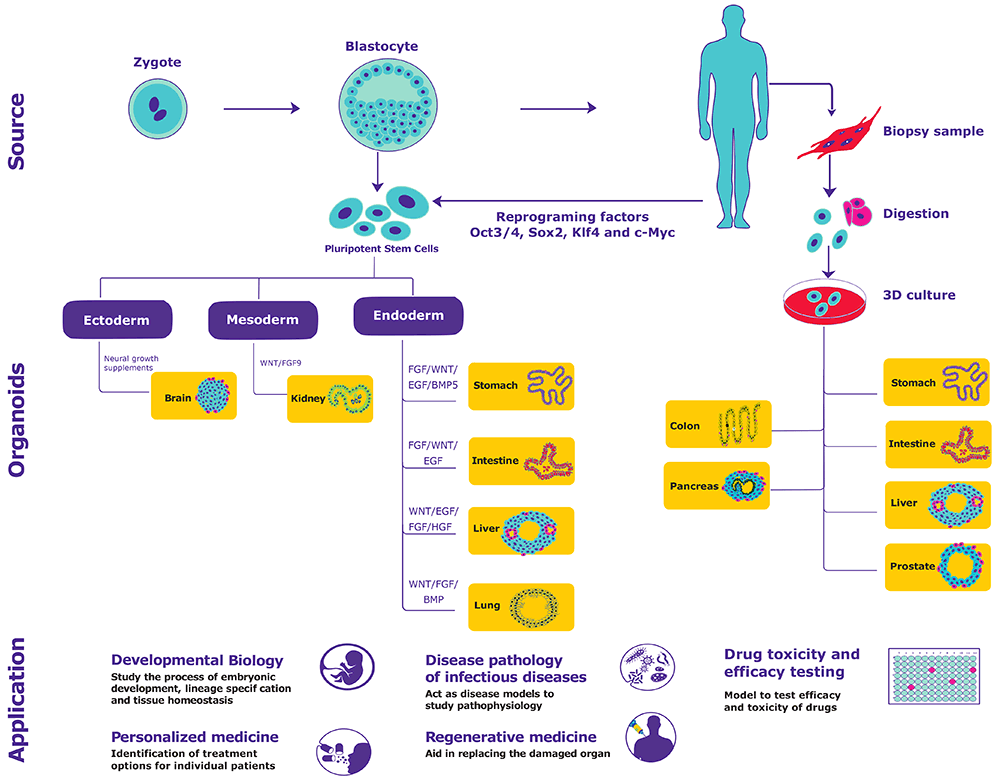
Model systems drive biological research by recapitulating body processes and functions from the molecular to whole organism level. The human body is composed of both cellular and non-cellular material organized in a highly specialized manner. It is difficult to mimic all aspects of human biology with one in vitro model system. 3D cell culture models are a more accurate representation of the natural environment experienced by cells in the living organism as opposed to growing cells on 2D flat surfaces.
What are Organoids?
Organoids are in-vitro derived 3D cell aggregates derived from primary tissue or stem cells that are capable of self-renewal, self-organization and exhibit organ functionality.3 Organoids address the limitations of existing model systems by providing:
- Similar composition and architecture to primary tissue: Organoids harbor small population of self-renewing stem cells that can differentiate into cells of all major cell lineages, with similar frequency as in physiological condition.
- Relevant models of in-vivo conditions: Organoids are more biologically relevant to any model system and are amenable to manipulate niche components and gene sequence.
- Stable system for extended cultivation: Organoids can be cryopreserved as biobanks and expanded indefinitely by leveraging self-renewal, differentiation capability of stem cell and intrinsic ability to self-organize.
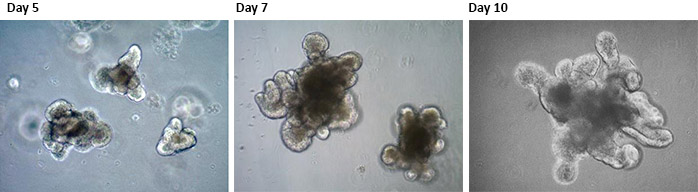
Figure 1.Mouse Intestinal Epithelial Organoids. 3D Organoids were generated from adult mouse intestinal tissue following the protocol outlined by Clevers et al. Science. 2013. Organoid cells begin to form lumens and bud structures at around day 3-5 in culture and form complex crypt-like structures around day 7-10. These crypt-like domains are functionally similar to those of the adult intestine, where dividing LGR5+ intestinal stem cells are intercalated with Paneth cells located at the crypt base.
Cell Lines
Organoids vs. Spheroids
Organoids and spheroids are both cells cultured in 3 dimensions. Spheroids are often formed from cancer cell lines or tumor biopsies as freely floating cell aggregates in ultra-low attachment plates whereas organoids are derived from tissue stem cells embedded within an ECM hydrogel matrix such as Matrigel. Organoids are highly complex and are more in vivo-like when compared to spheroids. Recently, tumor organoids have shown to predict how well patients respond to cancer drugs to aid in personalized medicine.
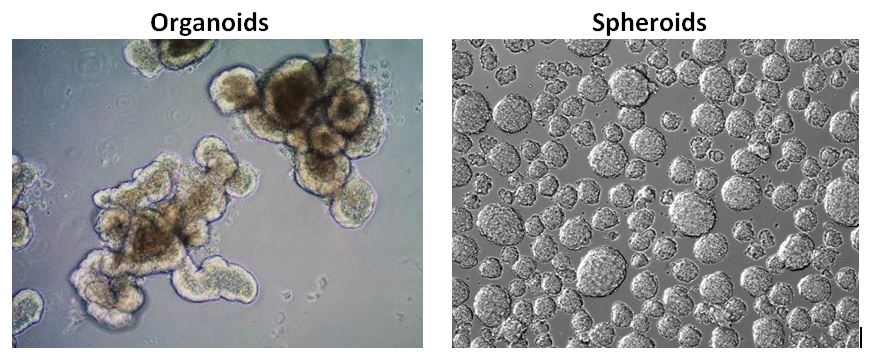
Figure 2.Organoids vs. Spheroids. Stem cell derived organoids have more in vivo-like phenotypes with higher order tissue complexity compared to tumor spheroids.
How are Organoids Generated?
Organoids are generated either from primary tissues or pluripotent stem cells (induced pluripotent stem cells (iPSC) or embryonic stem cells (ESCs)) by providing appropriate physical and biochemical cues4.
Physical cues: Provide support for cell attachment and survival. Examples include collagen, fibronectin, entactin and laminin.
Biochemical cues: Modulate signaling pathways, thereby influencing proliferation, differentiation and self-renewal. Examples include EGF, FGF10, HGF, R-spondin, WNT3A, Retinoic acid, GSK3β inhibitors, TGF-β inhibitors, HDAC inhibitors, ROCK inhibitors, Noggin, Activin A, p38 inhibitors and Gastrin.
Organoid Qualified Reagents
Applications of Organoids
Organoids are physiologically relevant and amenable to molecular and cell biological analyses, holding great promise in both basic research and translational applications.
Developmental Biology
Organoids derived from ESC, iPSCs retain features of their developmental stage and help in studying the process of embryonic development, lineage specification and tissue homeostasis. It also shed light on development of stem cells and their niche.
- Development of organs such as brain24, pancreas25 and stomach7 was studied through sequential differentiation steps inducted by modulating Wnt, BMP and FGF signaling pathways
Disease Pathology of Infectious Disease
Organoids represents all components of organ and are suited to study infectious diseases affecting specialized human cell types.
- Lung organoids derived from iPSCs from healthy child carrying null alleles of interferon regulatory factor- 7 gene employed to study influenza virus replication 26
- Forebrain organoids derived from human iPSC was employed to study infection of zika virus on neural progenitors27
Regenerative Medicine
Transplantation of organoids derived from the adult stem cells aid in replacing the damaged organ or tissue. In addition, feasibility for gene correction using CRISPR/Cas9 technology can be used in treating monogenic hereditary diseases.
- Small intestine organoids retained characteristics of small intestine, such as villus formation and presence of paneth when transplanted in mouse models28
Drug Toxicity and Efficacy Testing
The possibility to test efficacy and toxicity of drugs against representative targets/organs (gut, liver and kidney) could potentially limit the ethical issues associated with animal usage.
- Hyman kidney organoids were employed to demonstrate the nephrotoxicity of cisplatin 11
Personalized Medicine
Organoids derived from adult stem cell of individual patients allows ex-vivo testing of drug response.
- Colon organoids were employed to identify treatment options for patients with rare CFTR mutations29
- Tumor organoids can be employed to assess the drug response at the level of individual patients.