Improved LC-MS Analysis of Compounded Semaglutide Impurities Using a Positively Charged Surface (PCS) C18 Column
Abstract
The newest generation of GLP-1 agonists offers significant health advantages for patients, including weight reduction and type-2 diabetes. Recent supply chain disruptions have constrained the commercial availability of these therapeutics, creating an opportunity for compounding pharmacies to market their own formulations. Utilizing an LC-MS methodology developed for liraglutide with the BIOshell™ A160 Peptide PCS-C18 column, we show that a compounded version of semaglutide surpasses ANDA guidelines for peptide-related impurities.
Section Overview
Introduction
Glucagon-Like Peptide (GLP-1) drugs have been on the market for almost two decades, beginning with the launch of exenatide in 2005.1 Nevertheless, the sales and therapeutic benefits of these drugs have only recently experienced dramatic growth with the advent of the latest generation of treatments. Compounds such as semaglutide (Ozempic®; Novo Nordisk) and tirzepatide (Zepbound®; Eli Lilly and Co.) have been designed to enhance both potency and half-life, enabling weekly subcutaneous administration rather than the previously required daily injections.
Semaglutide represents an advancement over Novo Nordisk's earlier therapeutic, liraglutide, through the incorporation of an alpha-amino butyric acid (AiB) at position 2, which provides resistance to proteolytic degradation by dipeptidyl peptidase-4.2 Additionally, the fatty acid chain linker connected to the substituted lysine at position 26 has been modified to an 18-carbon di-acid, enhancing biodistribution.
Recent reports have highlighted supply chain shortages for these next-generation GLP-1 agonists, which have enabled compounding pharmacies to offer custom formulations for sale, bypassing certain FDA regulatory requirements.3 This situation poses risks for patients, as peptide-related impurities may be introduced into these compounded formulations without adequate evaluation of potential immunoreactivity, potency, and off-target effects. Current Abbreviated New Drug Application (ANDA) guidelines for synthetic peptide drug products specify that peptide-related impurities should not exceed 0.5% of the Full Length Product (FLP), with the justification that such impurities "would not be expected to affect the safety or effectiveness of the proposed generic synthetic peptide as compared to that of the RLD (reference listed drug)"4. Furthermore, the FDA guidance require that any "impurities between 0.1-0.5% must be identified, characterized, and justified for not affecting the safety and efficacy"5.
Impurities may also arise from improper handling and storage of peptide therapeutics. Oxidation and deamidation are examples of modifications that can affect peptide therapeutics. The distribution of products like semaglutide through compounding pharmacies raises concerns that these therapeutics may not be appropriately handled, potentially introducing impurities through this pathway that pose risks to patients.
In previous work, we demonstrated the effectiveness of the BIOshell™ A160 Peptide PCS-C18 columns for characterizing a Critical Quality Attribute (CQA) of liraglutide, where the N-terminal histidine can undergo cyclization when exposed to trace concentrations of formaldehyde.6 This CQA had also been documented when liraglutide was exposed to specific excipients.7 Since semaglutide also possesses an N-terminal histidine, we sought to determine whether a comparable modification could be detected. We verified that cyclization of the N-terminal histidine to form an imidazopyrimidine ring does indeed occur in semaglutide. We also acquired a sample of compounded semaglutide along with a sample of a commercial formulation (Ozempic®) for comparative analysis. Our findings revealed that the levels of N-terminal histidine modification in the compounded semaglutide surpass FDA ANDA guidelines, whereas the Ozempic® sample remains within acceptable limits. The data suggests that this modification occurs because of inadequate handling during the production of the compounded product, introducing avoidable risks to patients.
The growing complexity of these peptide therapeutics has made liquid chromatography with mass spectrometric detection (LC-MS) an indispensable characterization tool. However, the basic nature and high hydrophobicity of these compounds present challenges for analysis using conventional reversed-phase C18 chromatography columns. Strong ion-pairing agents such as trifluoroacetic acid are typically necessary to achieve satisfying chromatographic performance. Regrettably, these strong ion-pairing agents are generally incompatible with mass spectrometry due to substantial ion suppression effects. More recently, mixed bonded phases like the BIOshell™ A160 Peptide PCS-C18, which combines C18 with a ligand which is positively charged under acidic conditions, creating a positively charged surface (PCS), have demonstrated enhanced resolving power and peak capacity when using weaker, more MS-compatible ion-pairing agents such as formic acid.
In this study, we demonstrate how the BIOshell™ A160 Peptide PCS-C18, 15 cm x 2.1 mm I.D. column outperforms a standard C18 column in separating full-length semaglutide product from the N-terminal imidazopyrimidine ring impurity, and how this column can be routinely employed to monitor for this impurity in any GLP-1 therapeutic containing an N-terminal histidine.
Experimental
Research-grade semaglutide was purchased from Cayman Chemical (Ann Arbor, MI; item# 29969) and dissolved in 10 mM Tris, pH 8.0, to achieve a concentration of 0.2 mg/mL. The samples were portioned into aliquots and maintained at -20 °C. A syringe containing compounded semaglutide at 1 mg/mL concentration was provided by a prescribed patient. Likewise, an unopened Ozempic® injector pen containing semaglutide at 2.68 mg/mL concentration was provided by a prescribed patient. Both the compounded semaglutide and Ozempic® samples were diluted to 0.2 mg/mL using LC-MS-grade water. A Shimadzu Nexera X2 HPLC system (Columbia, MD) was interfaced with a Thermo Q-Exactive HF (Waltham, MA).
The two superficially porous particle (SPP) columns that were evaluated for LC-MS in this study under the conditions described in Table 1 were:
- BIOshell™ A160 Peptide PCS-C18
- BIOshell™ A160 Peptide ES-C18
Results and Discussion
Consistent with our earlier findings when solubilizing liraglutide in Tris-HCl buffer containing trace formaldehyde, we detected two peaks in the MS total ion chromatogram (TIC) for the research-grade semaglutide sample dissolved in 10 mM Tris-HCl at pH 8.0, as displayed in Figure 1. Peak 1 corresponds to the FLP semaglutide, while peak 2 corresponds to the +12 Da N-terminal histidine modification in Figure 1 and all subsequent figures. A comparable +12 Da mass shift in the deconvoluted spectra between peaks 1 and 2 is observed and presented in Table 2. The N-terminal modification was verified through MS/MS analysis and is not displayed.
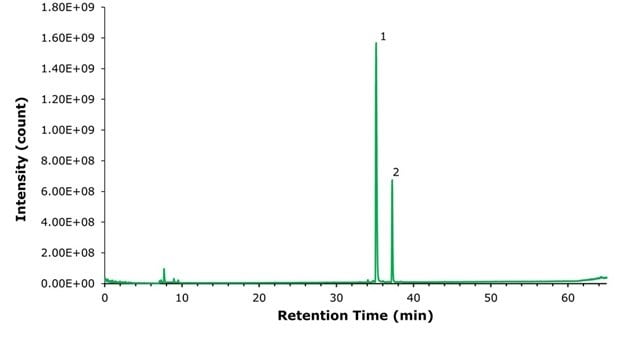
Figure 1.Chromatogram of 100 ng research-grade semaglutide. Peak identities described in Table 2.
A 100 ng sample of compounded semaglutide and 100 ng of commercial semaglutide (Ozempic®) were examined under identical analytical conditions. The TICs are shown in Figures 2 and 3 respectively, and Figure 4 presents a magnified comparison of the N-terminal histidine modification levels.
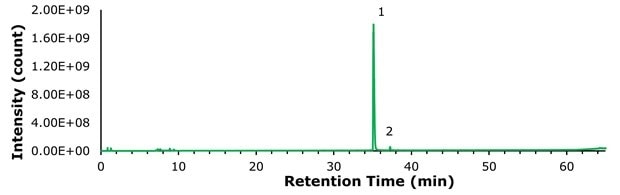
Figure 2.TIC of 100 ng compounded semaglutide.
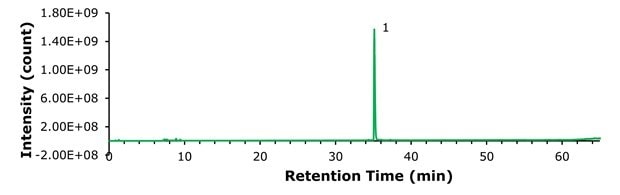
Figure 3.TIC of 100 ng commercial semaglutide (Ozempic®).
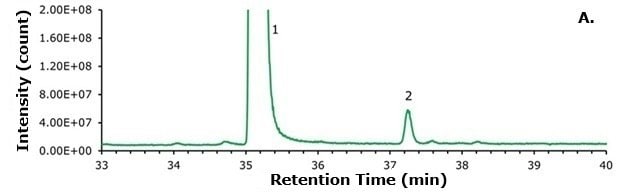
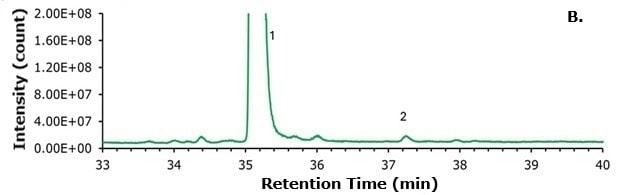
Figure 4.Zoomed-in comparison of (A) compounded semaglutide and the (B) commercial sample (Ozempic®). The compounded semaglutide sample shows a significant increase in the N-terminal histidine modification (Peak 2).
As shown in Figure 4, a significant amount of the N-terminal histidine modification is observable from the compounded sample. A much smaller amount is seen in the pharmaceutical grade (Ozempic®) sample. To assess the relative quantities of each analyte, two distinct methodologies were employed. The first method compared the abundance of deconvoluted peaks using the Thermo Freestyle software, with relative amounts calculated for each species, including identified Na+ and K+ adducts. The second method involved integrating the peaks in the TIC. The relative amounts are presented in Table 3.
As demonstrated in Table 3, the N-terminal histidine modification is present at levels 3-4 times higher in the compounded sample compared to the Ozempic® sample. This concentration exceeds FDA guidelines for permissible impurity levels in peptide pharmaceuticals and poses a risk to patients. It remains uncertain whether this specific modification has been evaluated with respect to potency and immunogenicity. However, it is likely evident that any existing GLP-1 agonist containing this N-terminal histidine is susceptible to this modification through inadequate manufacturing or handling practices. This likelihood includes liraglutide, semaglutide, dulaglutide, exenatide, lixisenatide, and potential therapeutics currently undergoing clinical trials at the time of this writing, such as amycretin.
To demonstrate the benefits of using a positively charged surface (PCS) C18 column in the presence of a weak ion pairing agent such as formic acid for LC-MS applications, we re-ran the semaglutide samples described above on a BIOshell™ A160 Peptide ES-C18 column with the same particle size, pore size, column dimensions, and methodology. This column is highly stable to acidic conditions (pH 1) but does not bear the mentioned positively charged ligand present in the PCS-C18. Figures 5, 6, and 7 show that on a standard C18 column the resolution declines while peak broadening and tailing increase considerably and would likely make a reliable identification of the N-terminal histidine modification much more challenging, even with high resolution MS.
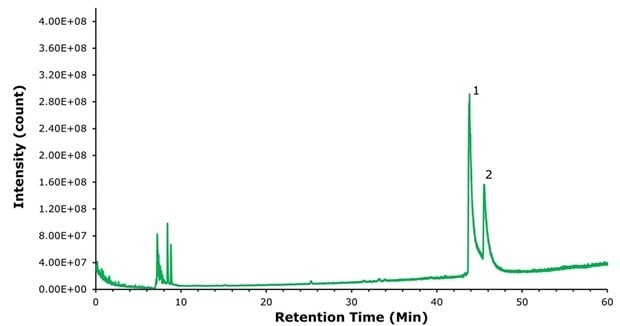
Figure 5.Analysis of research-grade semaglutide with BIOshell™ A160 Peptide ES-C18 column. The union of C18 with weak ion-pairing agent, formic acid, causes significant degradation of peak shape and resolution.
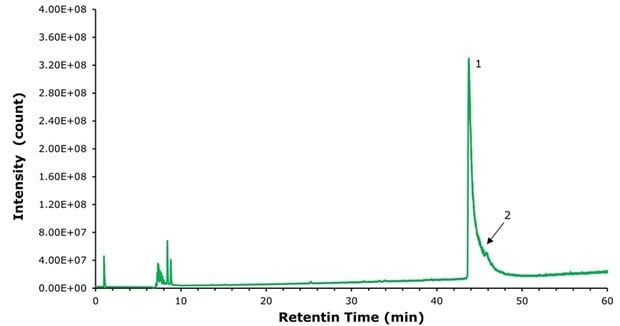
Figure 6.Analysis of Compounded semaglutide with BIOshell™ A160 Peptide ES-C18 column.
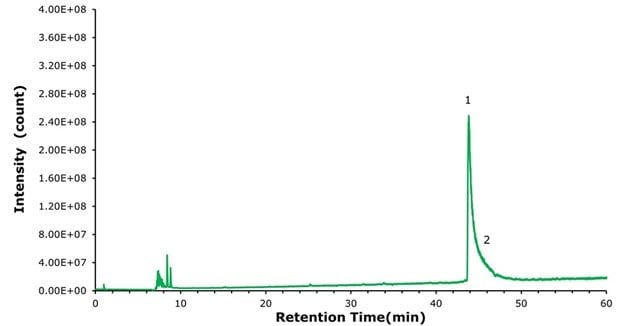
Figure 7.Analysis of Ozempic® with BIOshell™ A160 Peptide ES-C18 column. Peak tailing prevents detection of any N-terminal modification that was visible on the PCS-C18 column (Figure 4).
Conclusion
The BIOshell™ A160 Peptide PCS-C18 preserves excellent peak shape and resolution for basic compounds under weak ion-pairing conditions, enabling efficient impurity detection in various biopharmaceutical preparations. Utilizing the BIOshell™ A160 Peptide PCS-C18 column, an impurity in a compounded semaglutide sample could be detected, that surpasses FDA guidelines and a methodology was present that can be applied to screen additional compounded semaglutide samples for impurity-related risks.
Related Products
Solvents and Reagents
References
To continue reading please sign in or create an account.
Don't Have An Account?