Identification and Quantification of Liraglutide (GLP-1 Analogue) in Human Plasma
Abstract
A novel liquid chromatography-mass spectrometry (LC-MS) method was developed and validated for the quantification of liraglutide in human plasma. Liraglutide, a glucagon-like peptide-1 (GLP-1) receptor agonist, is widely used for the treatment of type 2 diabetes and chronic obesity. Sample preparation was performed by protein precipitation followed by solid-phase extraction using Supel™ Swift HLB SPE tubes to achieve efficient matrix clean-up. Chromatographic separation was carried out on a positively charged surface (PCS) BIOshell™ A160 Peptide PCS-C18 U/HPLC column. The method provided robust chromatographic performance and highly accurate detection in complex biological matrices.
Section Overview
Introduction
Liraglutide is a synthetic peptide primarily used for the treatment of type 2 diabetes and chronic obesity. The peptide belongs to the class of drugs known as glucagon-like peptide-1 (GLP-1) agonists.1 Liraglutide exerts its therapeutic effect by mimicking the activity of the endogenous hormone called GLP-1, which helps regulate blood glucose levels and appetite. Structurally, liraglutide consists of a 31 amino acid long peptide with a 16-carbon lipid side chain that enables albumin binding, thereby prolonging its half-life in plasma (Figure 1). Accurate evaluation of liraglutide concentrations in plasma is therefore important due to its protein binding and stability characteristics.
Human plasma represents a complex biological matrix containing high concentrations of proteins, which can lead to column clogging and ion suppression during LC-MS analysis. Therefore, it is important to perform suitable sample preparation.2 In this study, plasma proteins were first precipitated using an acetonitrile:water mixture, followed by centrifugation of the resulting suspension. Additional matrix cleanup was performed using solid-phase extraction (SPE) with Supel™ Swift HLB tubes. Chromatographic separation was carried out on a superficially porous particle (SPP, Fused-Core® technology) BIOshellTM A160 Peptide PCS-C18 U/HPLC column, which is designed to provide efficient separations of peptides and small proteins. The column incorporates a positively charged surface (PCS) that supports improved peak shapes and enhanced loading capacity for basic peptides, even when lower concentrations of weak-acid mobile phase modifiers are used.
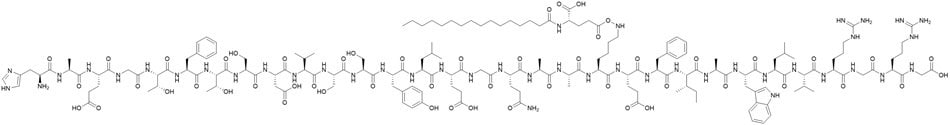
Figure 1.Liraglutide structure.
Experimental
Sample Preparation
Liraglutide spiked in human plasma
Liraglutide was dissolved in a 70:30 acetonitrile:water mixture to obtain a stock solution with a concentration of 1 mg/mL. The solution was further diluted with water containing 0.1% formic acid (FA) to prepare spiking solutions with concentrations of 2000ng/mL, 1000ng/mL, 800ng/mL, 600ng/mL, 400ng/mL, and 200ng/mL. The diluted solutions were added to human plasma in a 1:1 ratio )100 µL liraglutide spiking solution to 100 µL plasma(. The final concentrations in the SPE extract applied to LC-MS analysis are shown in Table 1.
To the plasma:liraglutide mixture (200 µL), 100 µL acetonitrile was added, resulting in the formation of a white precipitate. The mixture was centrifuged at 10,000 RPM for 10 min at 4 oC. The resulting supernatant was directly loaded onto conditioned SPE tubes.
SPE Cleanup
For SPE clean-up, Supel™ Swift HLB SPE tubes (1 mL/30 mg, 57493-U) were used in combination with a 24-port Visiprep™ vacuum manifold. The cartridges were conditioned with 500 µL water prior to loading the complete supernatant obtained from the centrifugation step described above. A wash step was performed using 500 µL water. Liraglutide was subsequently eluted using 2x 300 µL acetonitrile:methanol (70:30 v/v) containing 0.1% formic acid (FA).
LC-MS Method
Sample analysis by LC-MS was performed using a BIOshellTM A160 Peptide PCS-C18 column (2.7 µm, 150 x 2.1 mm I.D. 50659-U). Mobile phases containing 0.1% formic acid (FA) in the mobile phases were used for the chromatographic separation. The LC-MS conditions applied for the analysis of liraglutide in plasma extracts are summarized in Tables 2 and 3
Results and Discussion
LC Conditions
The structure of liraglutide, which contains both a hydrophobic palmitic side chain and a polar peptide backbone, presents challenges for chromatographic separation. The selection of a suitable stationary phase is therefore critical to achieve adequate retention and good peak shape. For this reason, a BIOshell™ Peptide PCS C18 column was employed on which liraglutide eluted at 5.3 min under the selected chromatographic conditions. The positively charged surface of the Fused-Core® particles consistently provided excellent peak shapes (Figure 2) and allowed for flexible injection volumes ranging from 1 to 10 µL (Figure 3).
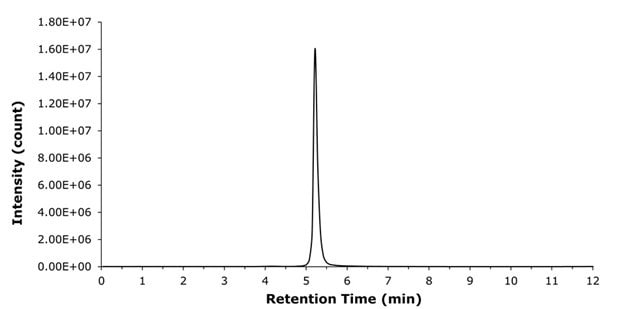
Figure 2.XIC (938.2468 u) chromatogram of liraglutide.
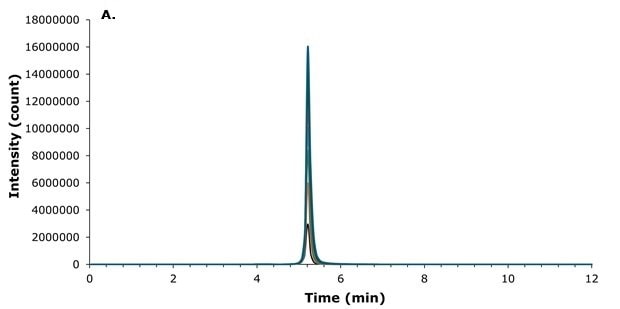
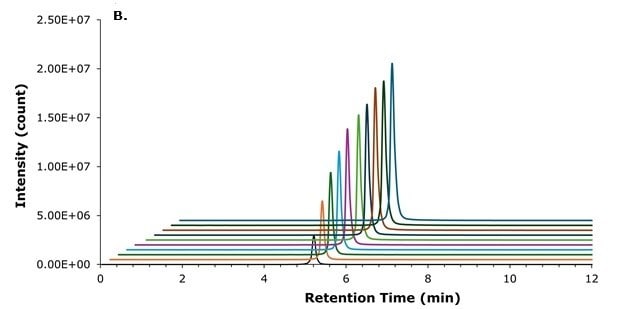
Figure 3.Loading study using injection volumes from 1 to 10 µL (133.3 ng/mL liraglutide extract solution) peak overlay without (A) and with (B) offset.
MS-Detection
Liraglutide, with an exact mass of 3748.9465 u (C172H265N43O51), was primarily detected by targeting its quadruply charged ion (z=4) at m/z 938.2468 u, which was observed to be one of the most abundant charge states alongside the triply charged ion (z=3). Furthermore, broadband collision-induced dissociation (bbCID) fragmentation patterns provided highly accurate confirmation, enabling confident identification of liraglutide even in this complex biological matrix (Figures 4 and 5).
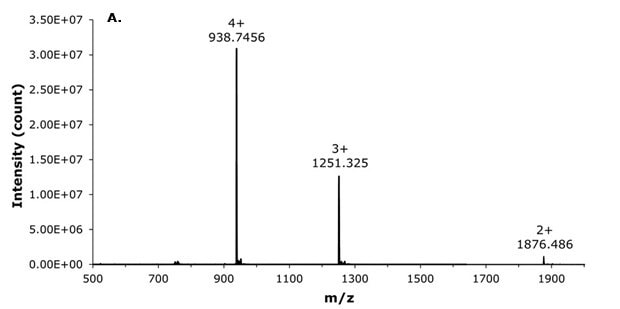
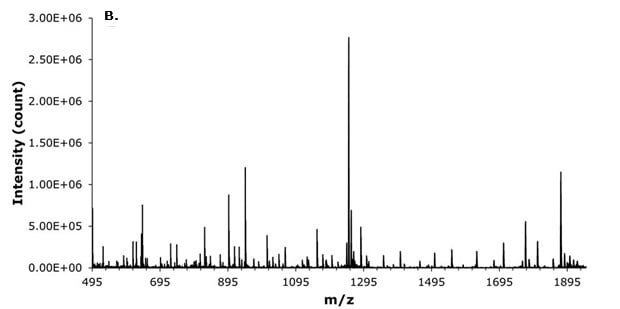
Figure 4.MS spectra of liraglutide masses for (A) MS z= 4, 3, and 2, and (B) MS/MS BBCID spectra.
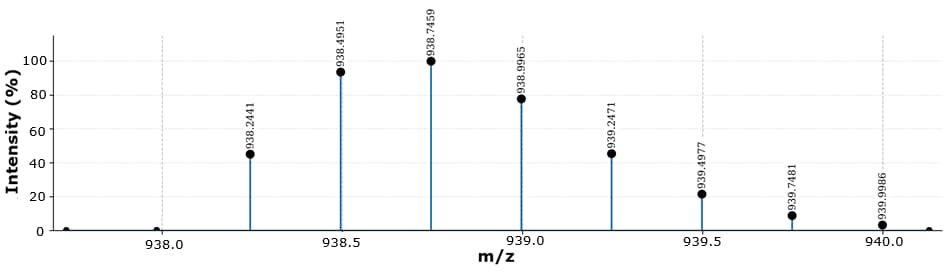
Figure 5.Isotopic pattern of liraglutide z=4 in the range of 938.00 to 940.00 m/z.
Purification of Spiked Liraglutide from Plasma Samples
To verify the LC-MS method and sample preparation procedure, liraglutide was spiked into human plasma samples. The spiked plasma was purified using a two-step protocol. First, proteins were precipitated by the addition of acetonitrile followed by centrifugation. Second, the resulting supernatant was further purified using Supel™ Swift HLB SPE tubes, which contain a hydrophilic-lipophilic-balanced polymer, and was then directly analysed by LC-MS. Using this procedure, the method sensitivity was determined with a limit of detection (LOD) of 26.62 ng/mL and a limit of quantification (LOQ) of 80.68 ng/mL for the injected solution, corresponding to plasma concentrations of 159.72 and 484,08 ng/mL, respectively. The recovery for the 66.67 ng/mL spike concentration was determined to be 58%.
The spiked samples were further used to generate a matrix-matched calibration curve for the determination of liraglutide in plasma, as shown in Figure 6.
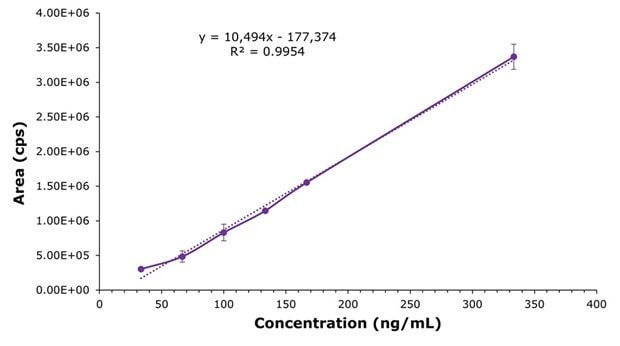
Figure 6.Matrix-matched calibration curve for liraglutide spiked plasma samples (33.33, 66.67, 100, 133.33, 166.67 and 333.33 ng/mL in the final SPE extract solution).
The analytical method provided effective separation of liraglutide from plasma proteins, enabling more accurate peak area determination and improved detection. Liraglutide eluted at a retention time of 5.3 min, while most matrix-related impurities eluted after 7 min (Figure 7). In the extracted ion chromatogram (XIC) of a liraglutide spiked sample, no inferences were detected (Figure 8).
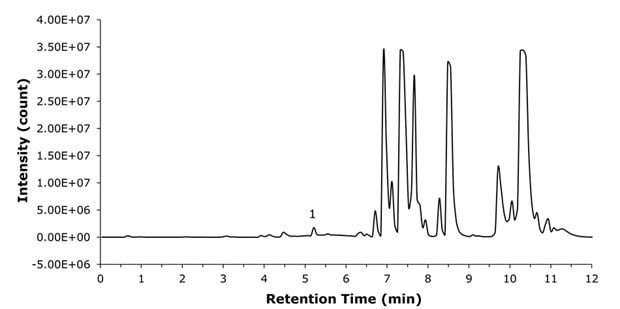
Figure 7.BPC chromatogram of spiked plasma sample. Liraglutide (1) elutes at 5.3 min.
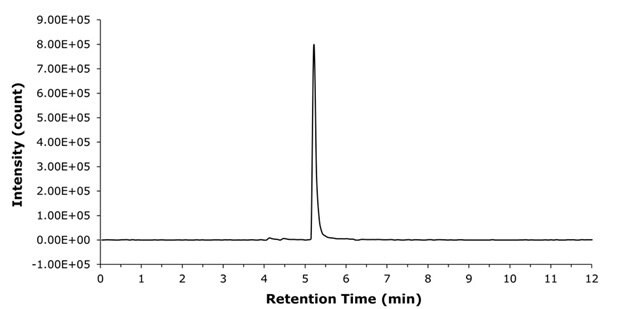
Figure 8.XIC (938.2468 u) chromatogram of liraglutide spiked in plasma sample.
Conclusion
In this study, an LC-MS method for the identification and quantification of liraglutide in human plasma samples was successfully developed. The method incorporates a streamlined purification protocol with protein precipitation followed by SPE clean up using Supel™ Swift HLB SPE tubes containing a hydrophilic–lipophilic balanced polymer sorbent. This approach provided efficient sample preparation and allowed the simultaneous processing of 18 samples using a 24-port manifold. The sample preparation approach also offers potential for further scale-up of sample throughput by automation using 96-well plate formats or dispersive in-pipette tip SPE (DPX).
A key strength of the method lies in its chromatographic performance, which achieved effective separation of liraglutide from complex plasma matrix components. This separation supports reliable detection and accurate quantification of liraglutide by minimizing potential matrix interferences. The straightforward and potentially high-throughput purification protocol, combined with the analytical precision of the LC-MS system employing an efficient Fused-Core® BIOshell™ A160 Peptide PCS-C18 U/HPLC column, makes the method particularly well-suited for routine bioanalytical applications.
Related Products
HPLC Column and SPE
Reagents and Solvents
References
To continue reading please sign in or create an account.
Don't Have An Account?