Process Sampling: Microbial Control and Risk Mitigation
Microbial control is a critical aspect of drug manufacturing and relies on a comprehensive strategy to eliminate or inhibit the growth of all microorganisms, including bacteria, fungi, and viruses, during processing. Common approaches include sterilization or sanitization of equipment or implementation of presterilized single-use technologies. Microbial contamination can occur at any point in processing and investigations into the cause or source of the contamination and identification of the contaminant are disruptive and costly to manufacturers and can delay the production of critical pharmaceuticals.1
Learn more about bioburden control and mitigation of microbial contaminations risks in biopharmaceutical manufacturing in this article.
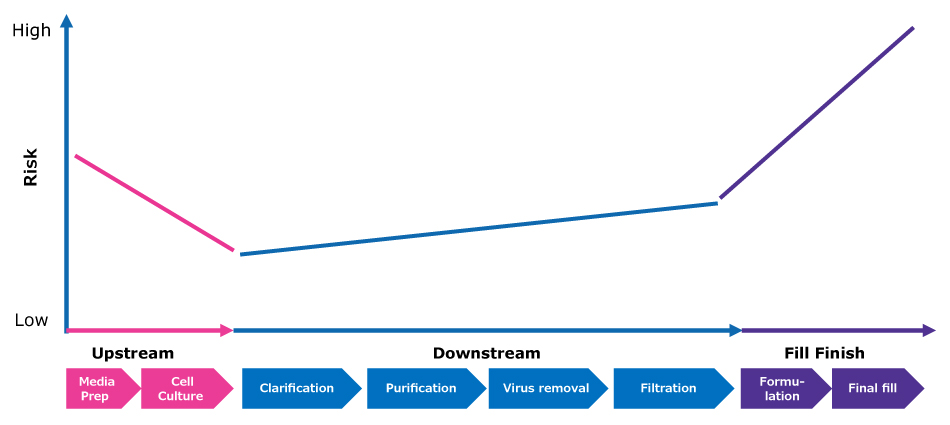
Figure 1.Graphical representation illustrating the risk dynamics throughout a biopharmaceutical process.
Intrinsic Risk and Regulatory Requirements for Bioburden Control in Biopharma
Drug manufacturing carries an intrinsic risk from microbial contamination and protecting any process from contamination relies on:
- Preventing microbial entry into the process through selection of high-quality raw materials and chemicals
- Robust sampling and sensitive testing to detect the presence of microbial contaminants
- Filtration technologies to control bioburden load.
Attention to each of these aspects of control is key for successful bioburden controlled and aseptic drug processing.
The updated EU GMP Annex 1 includes a requirement for a documented contamination control strategy (CCS) in the manufacture of sterile medicinal products.2 This CCS outlines the combination of approaches that enable aseptic processing and reinforces the importance of reliable methods for monitoring and controlling microbial contamination.3 PDA Technical Report No. 69 provides valuable insights on both process design and bioburden prevention considerations for pharma and biopharma manufacturers and highlights the importance of a comprehensive sampling strategy for managing bioburden in drug manufacturing.4
Is Bioburden Control the Same Throughout Processing?
Bioburden control in early processing steps is focused on the sourcing of high-quality raw materials to prevent the introduction of adventitious agents into the process. Contamination risk is especially high in upstream cell culture steps due to the nutrient-rich environment, which is ideal for microbial proliferation. Although contamination events in upstream cell culture steps rarely have serious implications for final product quality or patient safety, they are of great concern for manufacturers because they are disruptive and costly with significant implications for processing continuity and drug supply reliability.
By contrast, downstream biopharma purification operations operate as “low bioburden”, where bioburden is present but routinely monitored and controlled. As many process buffers and sanitizers are less hospitable to microbes than cell culture media, the risk of microbial proliferation is lower than in upstream, but it is not zero. Some steps involve components that are reused multiple times, such as chromatography resins and tangential flow filtration devices; these components cannot be sterilized and are sanitized to minimize bioburden. Although control tends to focus on limiting proliferation of bacteria or fungi, the consequence of proliferation in terms of microbial byproducts also needs to be considered as these can impact the quality of the drug being produced.
There is no strict guidance on bioburden levels in downstream operations and manufacturers typically set their own control levels. The BioPhorum Operations Group Bioburden Working Group reported that action levels are commonly set at 1 to 10 colony-forming units per mL.5 Later in downstream operations, stringent aseptic control is maintained during fill finish steps. Any contamination here could impact product quality and patient safety.
Effective bioburden control strategies rely on three complementary approaches: assessing bioburden, mitigating the risk of occurrence, and monitoring on an ongoing basis to assure process control. Risk assessment tools can evaluate all aspects the process to determine the potential impact of contaminants.5 This methodical approach to risk assessment is based on recommendations from the International Council for Harmonisation document, ICH Q9.6
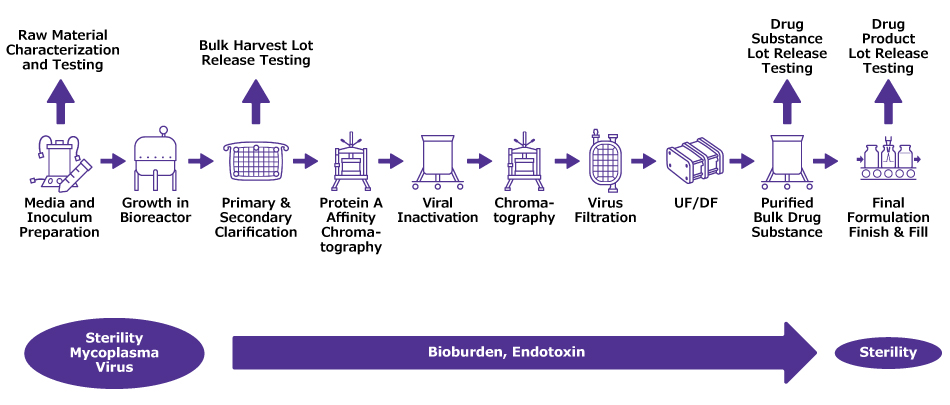
Figure 2.Idealized plan for routine bioburden and endotoxin monitoring throughout a downstream process. Raw materials and lot release testing will have additional tests.
Mitigate Contamination Risk in (Bio-)Pharmaceutical Manufacturing
Preventing microbial entry to pharmaceutical and biopharmaceutical production processes starts with the raw materials for production. Comprehensive information and data on the raw materials and components that confirms they meet the technical, regulatory, and supply needs for their designated application, use, and function is required during drug approval process. Availability of this type of documentation, accelerates risk assessments and helps you maintain compliance. For more information on the types of information explore our Emprove® Program.
Selecting the right raw materials and chemicals for each stage of development and manufacturing can minimize risk throughout your process. Our Emprove® Chemicals portfolio provides the high quality chemicals and raw materials, to help you from the early stages of pharmaceutical manufacturing to higher risk applications where low microbial and endotoxin levels are of the utmost importance and must meet compliance standards for drug production.
Find out more in our technical article “Parenteral drugs: Ensuring sterility and minimizing risks during manufacturing”.
Sanitizers and cleaning in place (CIP) solutions play an important role in any bioburden control strategy, particularly in processing steps where aseptic operations are not an option. Sanitization reduces, but does not eliminate, bioburden to an acceptable level. However, a disadvantage of sanitization is increased levels of microbial cellular debris, such as endotoxins, following treatment. For this reason, it is often beneficial to reduce bioburden by implementing filtration steps to remove potential contaminants, rather than relying on sanitization for microbial kill.
The increasing adoption of single-use technologies in drug manufacturing significantly reduces the likelihood of microbial ingress and mitigates the risk of biofilm formation as well as for sanitization or CIP chemical residues. These components, presterilized by irradiation or autoclave before use, offer clear advantages for microbial control. Similarly, closed sampling technologies offer advantages by minimizing the risk of introducing contaminants into the product flow path.
Maintain Control with Closed Sampling
Monitoring helps determine typical bioburden and endotoxin levels; where levels exceed a threshold limit, an investigation is usually required. In such cases, understanding the type of contaminant can help identify the source of the problem:
- Molds and spore formers such as bacillus often indicate environmental contamination
- Staphylococcus, Propionibacterium (or Cutibacterium) point to human contamination
- Non-fermenting gram-negative rods, such as Burkholderia or Ralstonia, suggest contaminated water systems or raw materials with high water content as the contamination source.
Even with contaminant identification, investigations still need to include detailed analysis of the system, materials, and operations to confidently establish route of ingress and appropriate corrective actions.
Risk assessments usually guide where samples are collected for routine monitoring, (Figure 2). Detailed plans define acceptable threshold levels, address how excursions will be investigated and evaluate potential impacts to the drug product safety profile.
Sampling for More Than Bioburden
In pharmaceutical and biopharmaceutical production, sample collection goes beyond bioburden analysis. Regardless of modality, unit operation, whether in preclinical development or commercial manufacturing, sampling is vital to monitoring critical process parameters (CPPs) and critical quality attributes (CQAs). As the industry shifts towards deeper process understanding and increased process control, through Process Analytical Technology (PAT), representative sampling methods are critical for assessing any process.
For additional information on sampling solutions, please visit Bioprocess Sampling Systems: Find the Right Sampling Approach for Your Needs.
