Optimizing mAb Capture using Multi Column Chromatography
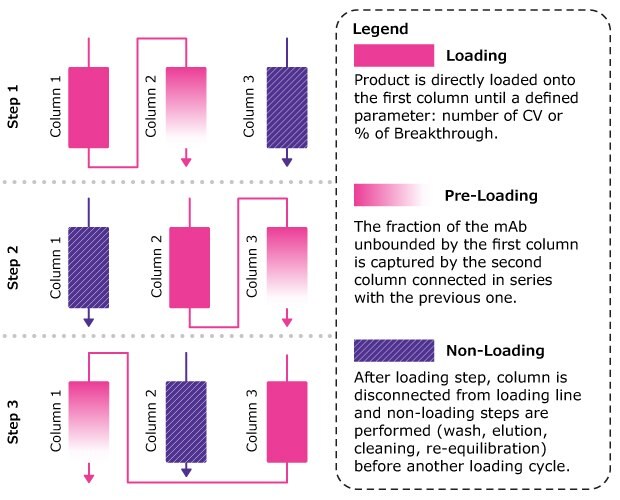
Figure 1.Multi column chromatography (MCC) workflow. MCC allows the simultaneous loading, wash, elution and regeneration of multiple columns.
Maximizing productivity of monoclonal antibody (mAbs) relies on integrating intensified processing into the workflow. The Protein A capture step can be a bottleneck in processing efficiency. However, shifting from a batch operation to continuous capture increases resin utilization through higher loading, while reducing the costs of resin storage and buffer consumption.
The Mobius® Multi Column Capture system was designed for continuous capture chromatography with single-use, closed technologies. This Technical Article summarizes an evaluation of the Mobius® Multi Column Capture system that assessed continuous mAb capture using multi column chromatography (MCC) approach, Figure 1. The study focused on: lab-scale process optimization using an ÄKTA Avant™ system, lab-scale process characterization of MCC with an ÄKTA Avant™ system and scale-up to 400 L with the Mobius® Multi Column Capture system.
For more information, please refer to our Application Note.
Discover more on how to intensify your downstream process—request a demo of the Mobius® Multi Column Capture system.
Lab-Scale Capture Process Optimization
Clarified, filter-sterilized harvest cell culture fluid (HCCF) contained the target mAb at 2.76 gmAb/L. The dynamic binding capacity (DBC) on Eshmuno® A resin was determined using a Vantage L11 column, packed with Eshmuno® A resin and qualified on an ÄKTA Avant™ system (Bed height (BH): 11.6 cm, Column Volume (CV) = 11 mL, Asymmetry (AS): 1.39, 2440 plates/m).
Figure 2 shows the DBC at 10% of the breakthrough: 28.6 g~mAb~/L~resin~ at 1 min residence time (RT), 39 g~mAb~/L~resin~ at 2 min RT, and 41 g~mAb~/L~resin~ at 4 min RT. A residence time of 2 min was selected given the small difference in productivity between 2 and 4 min RT.
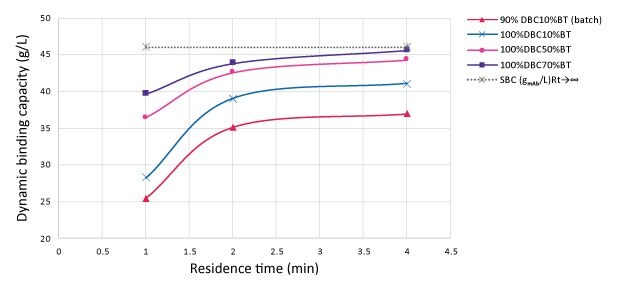
Figure 2.Influence of percent breakthrough on dynamic binding capacity as function of residence time
As the goal is to maximize resin use, breakthrough (BT) should also be considered. A 70% breakthrough at 2 min RT enables loading of 44 gmAb/Lresin resulting in resin utilization of 95%, against the 80% utilization for a batch process with 4 min RT.
| Resin utilization (%) = | Quantity of mAb retained (g) x Column Volume (L) |
| Static binding capacity (g/L) | |
The number of Column Volumes (CVs) corresponding to 70% breakthrough at 2 min RT was 16.5 CV, resulting in resin utilization of 95%. These parameters, defined on a single column, were then used for the MCC continuous operation. Table 1 lists the details of a single capture cycle. RT of non-loading steps was adjusted to improve continuity between loading and non-loading steps.
Lab-Scale Characterization of MCC
Lab-scale characterization simulated a multi column capture process using an ÄKTA Avant™ system coupled with single-use assemblies to enable continuous mode with three columns, reproducing the Mobius® Multi Column Capture system. This setup also included a reserve collection to recover unbound mAb, enabling reprocessing to improve yield and productivity.
To operate the ÄKTA Avant™ system continuously, buffers were diverted to selected columns with a valve. Sample was applied using a pump and diverted to the selected columns using an injection valve. Each column was connected to an inlet valve allowing diversion of the flow through to the next column, to waste, to reserve or to collection (elution). pH, conductivity and absorbance (UV280 nm) were monitored for the column in the non-loading phase only. The multi column operation was automated using Unicorn® 7.1 software.
The lab-scale parameters previously identified on a single column (70% BT, at 16.5 CV loading, 2 min RT) were used to process 1270 mL of HCCF at 2.76 g/L on the ÄKTA Avant™ system to simulate a multi column capture operation, Figure 3. There was a strong correlation between the lab-scale replicate runs of the MCC operation on the ÄKTA Avant™ system.
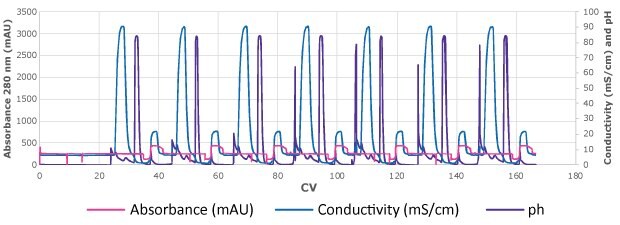
Figure 3.Chromatogram of lab-scale MCC set-up
Approximately 0.27 g mAb was detected in the reserve (first 1.1 CV of each wash step) which corresponded to 8% of the initial amount injected (3.51 gmAb). The 467 mL elution pool had a mAb concentration of 6.41 g/L (5 CV of collection, resulting in a combined process yield (reserve and eluate) of 95%. Aggregate levels in the final eluate pool were 2.32%.
Scale-up from Lab-Scale to the Mobius® Multi Column Capture System
Following confirmation of performance at lab-scale, large- scale MCC was performed using the Mobius® Multi Column Capture system. Three QuikScale® 200 columns packed with Eshmuno® A resin were used (BH: 6 cm, CV: 1.85 ±0.05 L, AS: 1.77 ±0.04, NETP: 3275 ± 1070 plates/m). Buffers, parameters and CV triggers determined during the lab-scale run were used in this large-scale trial.
The large-scale capture step trial with the Mobius® Multi Column Capture system using conditions and parameters determined at lab-scale (Table 2).
The overall, automated process required 8 hours to process 1.1 kg mAb (395 L of feed (2.8 g mAb/L resin)) over 12 complete and 2 partial loadings. A chromatogram of the run is shown in Figure 4.

Figure 4.Chromatogram of the Mobius® Multi Column Capture system showing absorbance at UV 308 nm after each column
Approximately 82.6 L of mAb at a concentration of 11.7 gmAb/L was recovered in the bag representing 966.4 g of product and 88.9% recovery. The aggregate level in the final eluate pool was 2.1%. The amount of mAb in reserve corresponded to 7.1% of initial product; if recovered, process yield would increase to ~94%.
In summary, 966.4 g of product was recovered in 8 hours of processing time with 5.6 L of total resin, resulting in productivity of 21.4 gmAb/L resin/h.
Process yield and high molecular weight aggregate levels were comparable between the lab- and larger-scale system as was the proportion of mAb recovered in the reserve. These data demonstrate that MCC operations can be effectively scaled from the lab-scale ÄKTA™ Avant system to the process scale Mobius® Multi Column Capture system, Table 3.
Benefits of Mobius® Multi Column Capture system for Intensified Processing
The Mobius® Multi Column Capture system is an automated, single-use solution designed to maximize the benefits of intensified capture of clarified harvest material from fed-batch or perfusion processes from 50 - 2000L in scale.
Improved Productivity: Using the protocol described in these studies, 395 L of clarified harvest material was processed in 8 hours (9 hours if the reserve was processed). By extrapolation, harvest material from a 2000 L bioreactor could be processed in 41 hours (or 46 hours if the reserve was processed). Productivity of the MCC operation was 21.4 gmAb/Lresin/h as compared to 8.6 gmAb/Lresin/h for a batch process.
Reduced costs: Implementing MCC with the Mobius® Multi Column Capture system reduces the volume of resin needed for purification as well as the volumes of buffers needed for purification; these reductions favorably impact the cost of goods. For the large- scale purification on the Mobius® Multi Column Capture system, 5.6 L of Eshmuno® A resin was used, as compared to 120 L required for a batch process of the same volume.
Enhanced productivity, reduced costs, and simplified scalability are compelling reasons for manufacturers to embrace the MCC approach for capture purification.
Related Products
To continue reading please sign in or create an account.
Don't Have An Account?