Microbial Environmental Monitoring
in the Pharmaceutical Industry
GMP Compliant Environmental Monitoring Solutions in Pharmaceutical Manufacturing
Because patient safety matters, the manufacture of pharmaceutical products is performed under strictly controlled conditions. Microbial monitoring is an important part of GMP regulatory compliance, used to prove that the manufacturing process is under control, especially in aseptic production.
Explore our broad portfolio of GMP-compliant solutions for microbial monitoring of aseptic pharmaceutical manufacturing including surface and personnel monitoring and microbial air monitoring. We also provide specific concepts for monitoring isolators and RABS to ease your workflows.
In addition to solutions for classical environmental monitoring, we have granulated as well as ready-to-use culture media for media fills/aseptic process simulations.
Learn more about our environmental monitoring portfolio and services for aseptic manufacturing within the brochure.

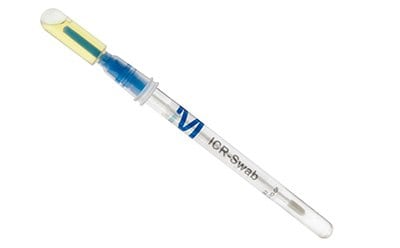
- ICR Swabs for surface and personnel monitoring in isolators and cleanrooms are designed for absence-presence testing on dry surfaces that are difficult to access.
- ICR Contact plates are designed to minimize the risk of contamination as they are produced in clean rooms. The ICRplus/RTplus versions are lockable and can be safely transported for incubation under aerobic, microaerophilic or anaerobic conditions.
- Hycon® Contact Slides are designed for monitoring flat and curved surfaces as well as personnel. The contact plates are available in standard single packaging or a double-bagged, gamma-irradiated format.

- The MAS-100 NT® and MAS-100 NT Ex® portable microbial air samplers are the industry standard for use in critical environments. These air samplers are available with a HEPA exhaust filter or can be upgraded with a filter mounting kit to reduce any risk of cross-contamination between low to high-grade cleanroom areas.
- The MAS-100 VF® active air sampler is specially developed for controlled environments. It is compact, easy to handle and ideal for monitoring the quality of your environment.
- Detect microbial contamination in gases with a few easy and automated steps. The MAS-100 Atmos® compressed gas sampler is simple and safe to use, and compliant with 21 CFR Part 11.
- ICR and ICRplus settle plates come with a high filling volume of 30 mL in 90 mm plates in order to reduce water loss during air monitoring. The plates are available in animal-free formulations to minimize the risk of BSE/TSE contamination.

Isolators and RABS are designed to prevent any human contact during controlled manufacturing. The MAS-100 Iso NT® & MAS-100 Iso MH® instruments in combination with the IsoBag® rapid transfer bag – allow a safe remote microbial air sampling for rapid culture media transfer into isolators.
Environmental monitoring in aseptic production isolators is made quicker by the IsoBag® as it provides ready-to-use gamma-irradiated contact or settle plates for immediate use. There is no need for decontamination cycles before introducing settle or contact plates, freeing up time for your actual production processes and thus saving costs.
Media Fills/Aseptic Process Simulation
Every aseptic process should be validated with at least 3 successful aseptic process simulations, or media fill tests. The aseptic process simulation should then be repeated regularly and the choice of the media for aseptic process simulation is crucial.
Granulated and ready-to-use culture media for secure media fill tests

With our irradiated, triple-wrapped granulated media or ready-to-use media you need not worry about compromising your validated process while performing media fill trials.
- Granulated culture media has been carefully formulated to ensure pharmaceutical manufacturers can perform efficient media fill trials with minimal risks.
- Ready-to-use culture media include Tryptic Soy Broth (TSB) and Vegetable Peptone Broth (VPB). These liquid broths are first sterilized by autoclave and then undergo a two-step pre-filtration process.
Watch Our Environmental Monitoring Webinars
Get your Aseptic Facility Ready for EU GMP Annex 1
The upcoming Annex 1 revision will include guidance specifically on designing a risk-based and effective contamination control strategy (CCS) based on a scientific assessment to understand the process and to apply risk management principles.
Watch WebinarAseptic Isolator Manufacturing: Introduction to Innovative Technology Solutions
Environmental monitoring within an isolator should be based on a formal risk assessment concerning the product, process and routes of entry. Watch the webinar to learn about regulatory requirements for environmental monitoring. Also, you will be introduced to innovative and effective technology solutions available to enhance your environmental monitoring program.
Watch WebinarHow Crucial is Culture Media Selection in Environmental Monitoring? A Roundtable Discussion
The choice of culture media plays a crucial role in quality control strategy in aseptic manufacturing for environmental monitoring in cleanrooms and isolators. Watch the webinar if you have faced challenges in your environmental monitoring practices and are interested to learn the impact of manufacturer requirements and design specifications of culture media in your current practices.
Watch Webinar
Environmental Monitoring Services
All our portfolios are supported by a broad range of services, embodying our ambition to remain one step ahead of customer demands and expectations. Benefit from our all-around services offers, performed by certified service engineers, that cover:
- Validation protocols and on-site IQ/OQ service
- Training
- Maintenance and service plans
- Traceability and calibration accuracy
Discover our Environmental Monitoring Solution
Contact our experts to request more information on environmental monitoring solutions for the pharmaceutical industry.
Related Articles
We have extensive knowledge about the workflow of environmental monitoring in pharmaceutical manufacturing areas. We fulfil all requirements requested for microbiological methods in the various GMP guidance’s as well as related in norms such as ISO 14698, EN 17141 and ISO 14644 by proven documentation.
- IsoBag™ ease the environmental monitoring workflow in aseptic production isolator
- Settle plates for isolators and critical cleanrooms
- Contact Plates and Swabs for Personnel and Surface Monitoring in Cleanrooms and Isolators
- High quality granulated and ready-to-use culture media irradiated for accurate and reliable aseptic process simulations.
- Microbial monitoring; environmental monitoring; interim storage; ICR contact plates; contact plates; TSA w. LTHThio Contact ICR+
Highlights
In this webinar, our media fill experts give an overview of what to consider when selecting media for aseptic process simulations.
This webinar outlines a holistic approach to ensure continued compliance, taking into account the expected changes with EU GMP Annex 1 (Draft 2020), including the need for EM programs and procedures based on risk analysis.
EN 17141, a new European standard for biocontamination control, has recently come into effect. In Europe, it will replace ISO 14698 Parts 1 and 2 from 2003, the previous standard for biocontamination control in cleanrooms and associated controlled environments.
In this 10-minute quick learning tutorial our expert Adèle Gisselmann will guide you through the requirements for aseptic manufacturers when selecting media for media fill tests or aseptic process simulations.
To continue reading please sign in or create an account.
Don't Have An Account?