Buchwald G6 Precatalysts: Oxidative Addition Complexes for Efficient L–Pd(0) Generation
The efficient generation of L–Pd(0) active species is critical to the development of efficient and robust cross-coupling reactions.1 Therefore, precatalysts with a pre-associated ligand to the metal center typically affords higher activity, shorter reaction time, and lower catalyst loading than a similar catalyst system which requires in-situ coordination of a ligand to the metal center.
Buchwald and coworkers have developed a family of Pd(0) precatalysts based on Pd(II) palladacycles with N,C-chelating ligands (Figure 1). These precatalysts are comprised of five generations (G1 through G5), which are distinguished by structural variations to the N,C-chelating ligand, and/or the anion X. Although the activation mode is the same for each precatalyst generation, deprotonation of the nitrogen leads to reductive elimination and generation of L–Pd(0). Each generation has unique advantages and disadvantages, which are described in previously published Technology Spotlights.2,3
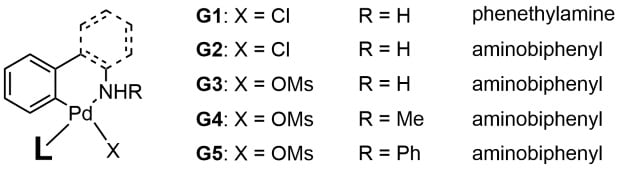
Figure 1.General Structures of Buchwald Precatalysts G1 to G5 (L = ligand)
The Buchwald G6 precatalysts are oxidative addition complexes (OACs), which exhibit the same advantages as the previous generations of Buchwald precatalysts: quantitative generation of L–Pd(0), thermal stability, air stability, moisture stability, ease of handling, and high efficiency (Figure 2).4 Furthermore, G6 Buchwald precatalysts demonstrate several comparative advantages over the previous generations of Buchwald precatalysts.
Key Benefits of Buchwald G6 Oxidative Addition Precatalysts for Pd-catalyzed Cross-coupling
- Catalyst activation does not require base and generates innocuous byproducts.
- OAC precatalysts are “on-cycle” intermediates that typically have higher reactivity and selectivity.
- Precatalyst synthesis is performed in a single step at room temperature.
- Versatile and tunable precatalyst design:
- Each of the three ligand types (X, L, and Ar) can be independently tuned to create a nearly endless number of precatalyst variations
- Improved solubility, greater stability, increased reactivity, and/or easier purification can be achieved by design or selection of X, L, and Ar
- Bulky ligands (e.g., tBuBrettPhos, AdBrettPhos, and AlPhos) are easily accommodated by G6 precatalysts

Figure 2.General structures and catalog number examples of three Buchwald G6 precatalysts.
Broad Reaction Scope and Applications of Buchwald G6 Palladium Precatalysts
Buchwald G6 precatalysts and other OACs have been applied as effective catalysts for the formation of C–C, C–N, C–O, C–F, and C–S bonds.4-9 Screening and comparison studies of a variety of catalyst systems and precatalysts typically show that OAC precatalysts have superior reactivity, selectivity, reaction scope, and/or yields.
Table 1. Reactions and Applications
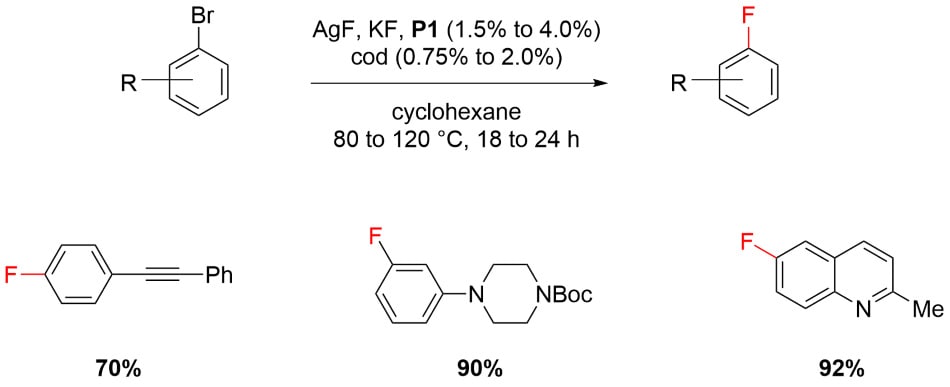
Fluorination of Aryl Bromides4
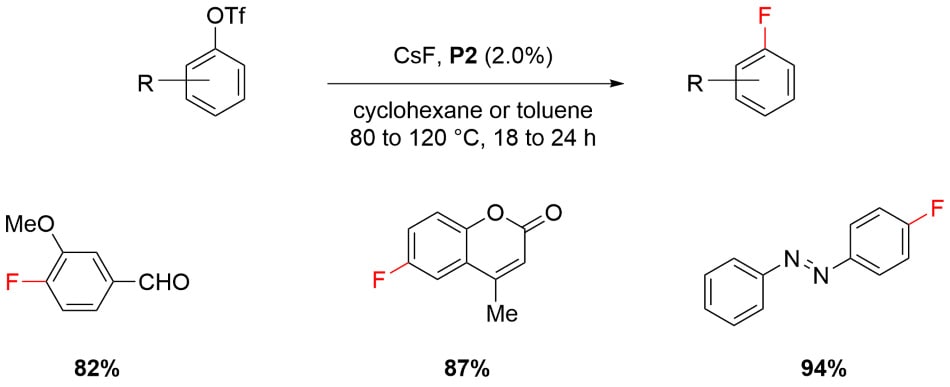
Fluorination of Aryl Triflates4
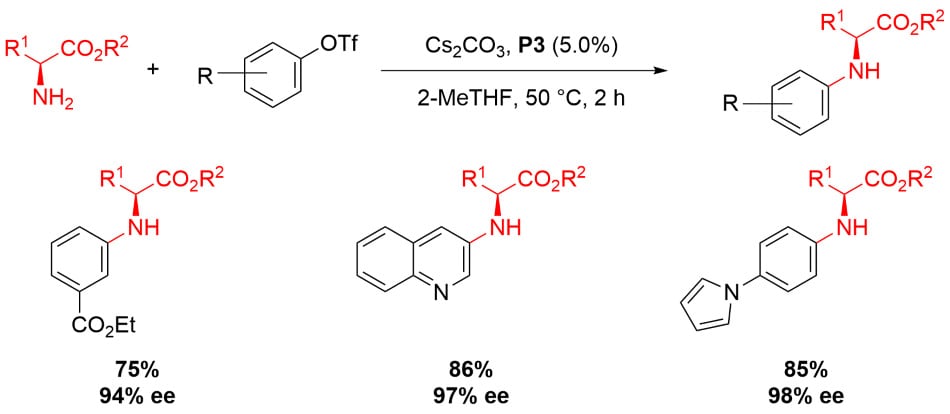
Amino Acid Ester Arylation4
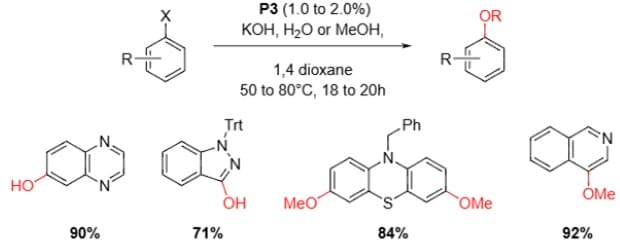
Alcohol and Hydroxide Coupling4
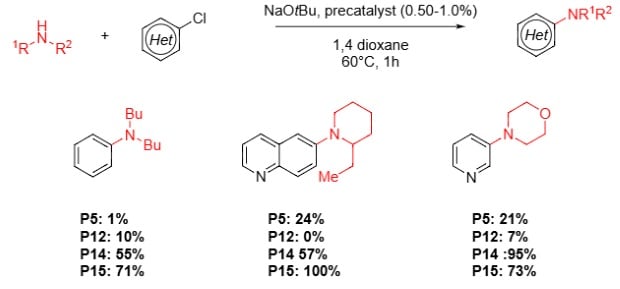
Buchwald Hartwig Aminations with Primary Aliphatic Amines7
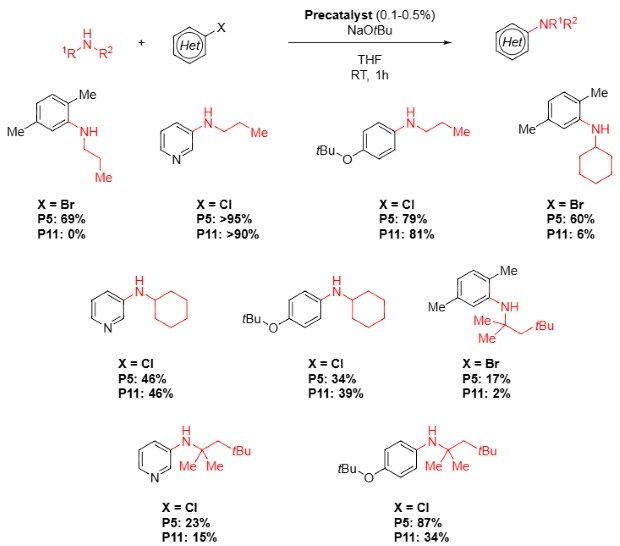
Buchwald Hartwig Amination with Alkyl Amines and N Heterocycles10
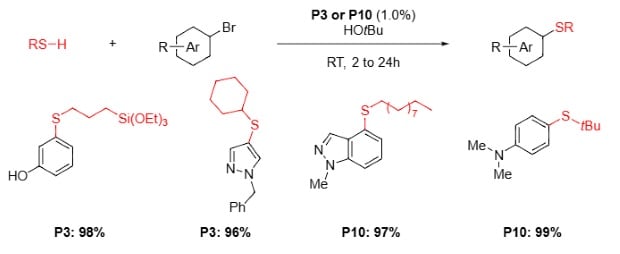
Aliphatic Thiol Coupling of Hetero(Aryl) Bromides9
Buchwald-Hartwig Amination of Base-sensitive Five-membered Heteroaryl Halides and Aliphatic Amines11
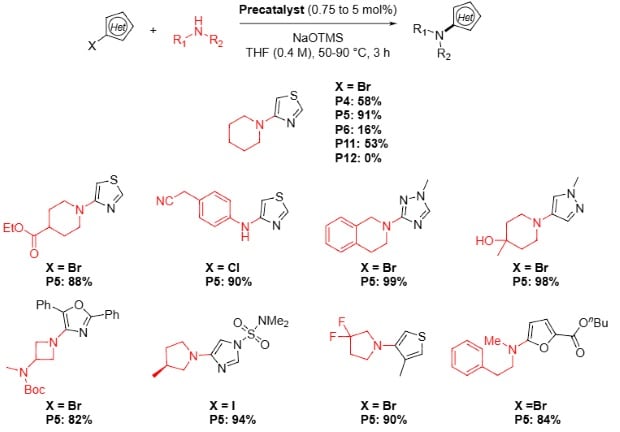
Table 2. Buchwald G6 and other OAC Precatalysts
For reaction and application details, see supplemental data sheet: Buchwald G6 Precatalysts: Oxidative Addition Complexes for Efficient L–Pd(0) Generation
References
To continue reading please sign in or create an account.
Don't Have An Account?