Protected Carbohydrate Building Blocks for More Efficient Syntheses
Introduction to Carbohydrate Chemistry
Carbohydrates and their biologically active glycoconjugate analogs are emerging as an important class of biomolecules. Despite the growing field of glycobiology, access to structurally well-defined oligosaccharides remains a significant challenge to chemistry, biochemistry, and chemical biology groups throughout the world (Werz 2012). One key challenge in oligosaccharide synthesis differentiating them from their protein and DNA analogues is the tedious multistep synthesis required to prepare a building block for further elaboration (Wang & Demchenko 2019). MilliporeSigma is proud to enable the carbohydrate scientists of the future to drive forward the field of glycobiology by enabling quicker access to well-defined glycoconjugates.
Advantages of Protected Glycoconjugate Analogs
There are a few, very common early stage protection strategies that are frequently employed to begin oligosaccharide synthesis. These strategies typically involve multiple steps to differentiate just a single hydroxyl on the carbohydrate building block. Further to multiple steps, some reactions tend to be low yielding further driving the need for repeated preparation.
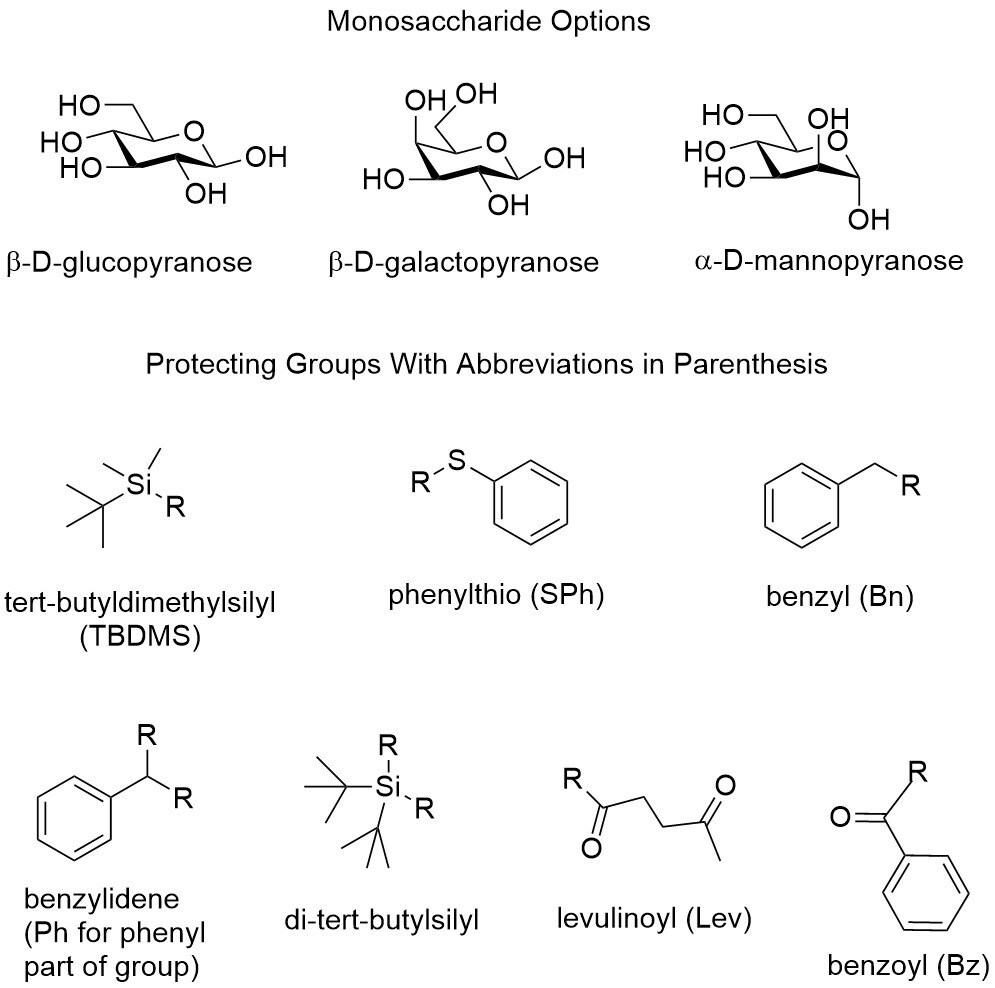
We are now offering a series of protected glucopyranose, galactopyranose, and mannopyranose building blocks. The above table shows the available monosaccharides and protecting groups. In all products, the hydroxy group on the anomeric carbon is protected with a phenylthio. For the other hydroxy groups, we offer combinations of the following protecting groups: 6-tert-butyldimethylsilyl; 6-benzyl; 4,6-benzylidene; 4,6-di-tert-butylsilyl; 3-levulinoyl; 2 and 3 benzoyl; and 2 and 3 benzyl. MilliporeSigma offers savings in time, reagents, and starting materials through the sale of these carbohydrate building blocks.
Chemical synthesis of carbohydrates and their surface immobilization: a brief introduction.
Werz DB.
Methods Mol Biol. 2012; 808:13-29. doi: 10.1007/978-1-61779-373-8_2. Review.
PMID: 22057515
Synthesis of carbohydrate building blocks via regioselective uniform protection/deprotection strategies.
Wang T, Demchenko AV.
Org Biomol Chem. 2019 May 28;17(20):4934-4950. doi: 10.1039/c9ob00573k. Epub 2019 May 2.
PMID: 31044205
Materials Table
如要继续阅读,请登录或创建帐户。
暂无帐户?