LC-MS/MS Analysis of Liraglutide on BIOshell™ A160 Peptide PCS-C18 Column
Abstract
In late 2024, the glucagon-like peptide-1 (GLP-1) agonist liraglutide attained generic status. Due to its distinctive characteristics, analyzing liraglutide through HPLC and LC/MS techniques presents significant challenges.
We present an LC-MS/MS methodology utilizing the BIOshell™ A160 Peptide PCS-C18 column for characterizing liraglutide samples, which successfully identifies multiple impurities, including an N-terminal cyclization that can occur even when the compound is handled under mild conditions.
Section Overview
Introduction
Glucagon-Like Peptide-1 (GLP-1) receptor agonists have shown extraordinary efficacy in treating conditions including type-2 diabetes and obesity, prompting pharmaceutical companies to accelerate development efforts aimed at enhancing both the potency and half-life of these therapeutic agents.1
Novo Nordisk introduced liraglutide in 2010 under the trade name Victoza®. The structure of liraglutide varies from the natural GLP-1 peptide through a single amino acid modification at position 34, where arginine replaces lysine. Furthermore, position 26 features an amide bond to the lysine side chain that connects a C16 fatty acid moiety (Figure 1). These structural alterations extend the half-life from roughly 10 minutes for native GLP-1 to 13 hours, thereby enhancing the drug's binding affinity to albumin and optimizing its biodistribution.
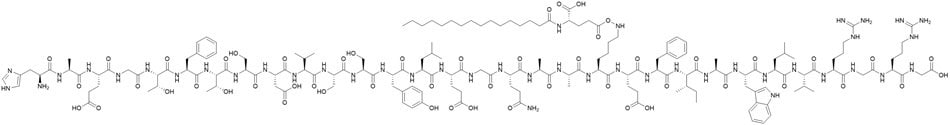
Figure 1.Liraglutide structure.
Liraglutide shares considerable structural similarity with glucagon, a generic peptide therapeutic routinely used to trigger hepatic glucose release from glycogen stores.
The US Pharmacopeia monograph outlines the quality control procedures for glucagon manufacturing, offering a valuable framework for QA characterization of liraglutide.2 Like glucagon, liraglutide is generally produced through recombinant expression in yeast, followed by acylation to attach the C16 fatty acid, with purification preceding QA analysis of the final liraglutide product. Nevertheless, the HPLC methodology detailed in the glucagon monograph employs a phosphate buffer at pH 2.7 on an L1 (C18) column with a 45-minute runtime, rendering it unsuitable for LC-MS characterization— a capability that will likely be essential for thorough liraglutide production QC. Conventional, strong ion-pairing reagents like trifluoroacetic acid (TFA) or phosphate-buffered mobile phases present compatibility challenges with mass spectrometry due to ion suppression effects or instrument contamination concerns. Consequently, when MS detection is required for characterization, weaker ion-pairing agents/buffers such as formic acid must be employed. While formic acid minimizes the risk of analyte ion suppression, it often compromises separation quality for basic compounds on conventional C18 columns, resulting in peak tailing. For the increasingly sophisticated QA analysis of biologics, stronger ion-pairing agents may offer advantages for standard HPLC-UV analysis; however, as mentioned, method modifications become necessary when MS characterization is needed. Optimally, a unified method suitable for both UV and MS detection, for QA and characterization would be advantageous.
BIOshell™ A160 Peptide PCS-C18 columns feature a positively charged surface (PCS), reversed-phase stationary phase engineered to deliver superior separations of basic compounds such as peptides and proteins when used with weak ion-pairing agents like formic acid. This attribute enables the development of an LC-MS compatible separation method while maintaining peak capacity.
The PCS-C18 platform facilitates implementation of a single separation approach applicable to both HPLC-UV detection for QA purposes and LC-MS characterization, thereby improving confidence in impurity identification and monitoring in a simplified method set up.
To illustrate the resolving power of the BIOshell™ A160 Peptide PCS-C18 phase using weak ion-pairing agents, Figure 2 displays an analysis of chemically-stressed glucagon. The method achieves baseline resolution of all four deamidation products, verified through LC-MS/MS analysis, within a 12.5-minute run. This method represents approximately a 75% reduction in method runtime relative to the procedure outlined in the current USP monograph for glucagon.
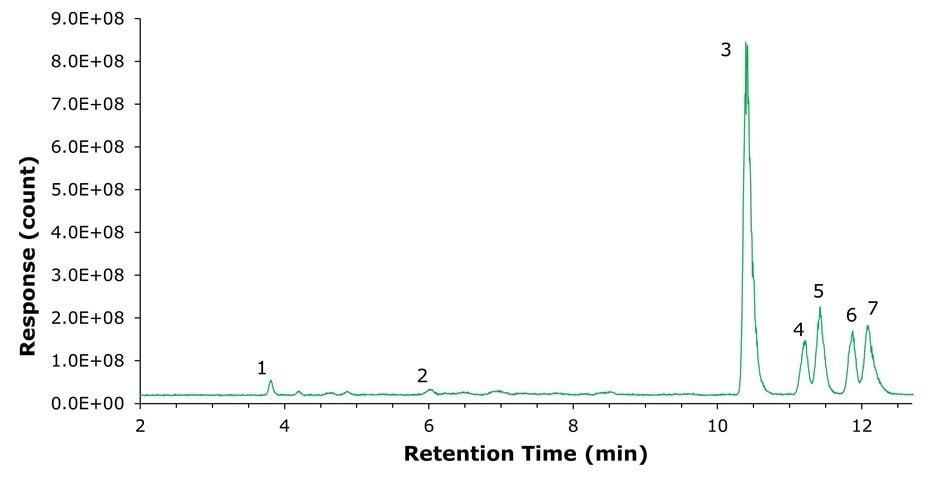
Figure 2.Separation of chemically stressed Glucagon.
Chromatographic Conditions: Column: BIOshell™ A160 Peptide PCS-C18, 2.7 µm, 15 cm x 2.1 mm I.D.; Mobile Phase: [A] Water (0.1% formic acid);
[B] acetonitrile (0.1% formic acid); Gradient: 20% B to 25% in 15 min, 25% B to 80% B in 0.5 min, hold at 80% B for 1.5 min; Detector: MSD, ESI - (+); Injection: 1.0 µL; Sample: Stressed glucagon, 100 µg/mL, water (0.1% formic acid).
Peak Identities: (1) Glucagon fragment, (2) Glucagon fragment, (3) Glucagon, (4) Asp28-glucagon, (5) Glu3-glucagon, (6) Glu20-glucagon, (7) Glu24-glucagon
In this investigation, we modified the analytical approaches used above for glucagon for the analysis of liraglutide using the BIOshell™ A160 Peptide PCS-C18 column. We successfully identified several potential impurities, including an N-terminal cyclization that appears to occur through base-catalyzed mechanisms in the presence of trace formaldehyde concentrations. The BIOshell™ PCS-C18 column enables efficient separation of these impurities from the main analyte when formic acid is used and ensures compatibility with both HPLC-UV and LC-MS/MS analytical workflows.
Experimental
Research grade liraglutide was dissolved in 10 mM Tris, pH 8.0, at a concentration of 0.2 mg/mL. Samples were aliquoted and frozen at -20 °C. A Shimadzu Nexera X2 HPLC system was coupled to a Thermo Q-Exactive HF.
The separation with MSD and UV detection was performed on a 15 cm x 2.1 mm I.D. BIOshell™ A160 Peptide PCS-C18 column with 2.7 µm superficially porous particles (SPP) under conditions listed in Table 1.
Results and Discussion
Preliminary analysis of a 500 ng liraglutide injection employing a long gradient method reveals two major peaks in the total ion chromatogram (TIC). The main peak appearing at 43.82 min corresponds to native liraglutide, which elutes at approximately 40% acetonitrile. A second, unanticipated peak attributed to a possible impurity is observed at 46.06 min. Figure 3 displays the +3 charge (z=3) state for both the primary and secondary peaks.
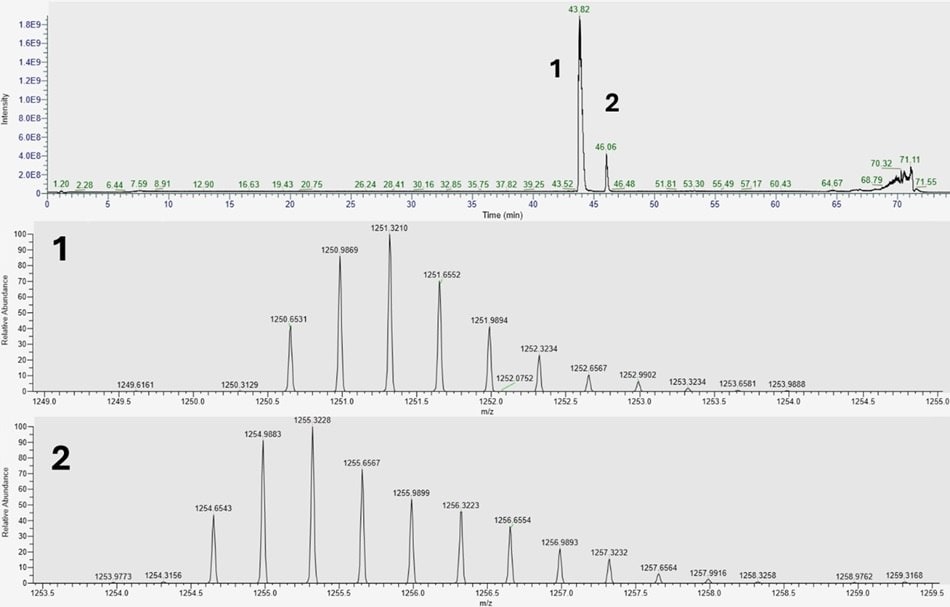
Figure 3.Long gradient results on the analysis of liraglutide. 1. Shows the 3+ charge state of liraglutide. 2. Represents an impurity of exactly +12 Da shift in absolute monoisotopic mass.
Deconvolution analysis of the 3+ charge states indicates that this impurity exhibits a mass increase of exactly +12 Da. MS/MS data pinpointed its location to the N-terminus. A review of the literature suggests this results from cyclization of the N-terminal histidine when exposed to formaldehyde.3 Further studies examining the effects of excipients on liraglutide modifications indicate that this N-terminal histidine demonstrates high reactivity to formaldehyde contaminants.4 We hypothesize that the Tris buffer used to dissolve liraglutide contained trace amounts of formaldehyde, leading to formation of the cyclized impurity.
A buffer with a pH range of 7.5-8.5 is required to solubilize most GLP-1 peptides due to their poor solubility under more acidic conditions. Likewise, most detergents and chaotropic agents are not compatible with MS methodologies. Even with our cautious approach to solubilization and storage, cyclization still took place. It may be advisable to incorporate an additive into the solution to capture trace formaldehyde.
The identified impurities detected in the research-grade liraglutide sample are listed in Table 2.
The identification of these impurities plays a critical role in pharmaceutical production to guarantee product purity and safety. Standard practice involves characterizing and documenting impurities present at levels as low as 0.1% relative to the main product.5
The here detected impurities, outside of the highlighted N-terminal cyclization impurity, are in such low concentrations that according to the USP guidance, they would not exceed the threshold of a to be monitored impurity as they would not affect the efficacy of the drug. Through comprehensive LC-MS characterization, manufacturers establish a reference standard that can be consulted during QA procedures when impurities emerge. This reference enables improved tracking of impurities within specific production lots and assists in determining the impurity's origin. The N-terminal cyclization impurity serves as an ideal illustration of this principle. This impurity’s presence could signal that formaldehyde contamination occurred at some point during the manufacturing workflow.
Since the BIOshell™ A160 Peptide PCS-C18 column enables, for basic analytes, excellent peak shape when using "weaker" ion-pairing agents like formic acid, faster methods with more sample throughput can be implemented. Figure 4 presents a UV trace at 280 nm from a 12-minute method that achieves baseline resolution between liraglutide and its N-terminal impurity in 0.1% formic acid. With more traditional C18 columns, achieving this level of separation would likely require the use of MS-incompatible ion-pairing agents such as TFA.
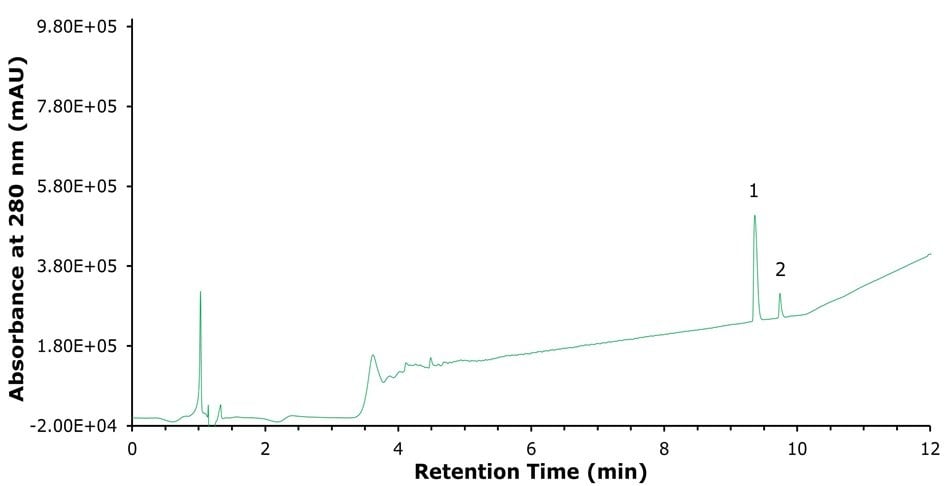
Figure 4.280 nm UV trace of a fast separation of 1) liraglutide and 2) N-terminal cyclization impurity on BIOshell™ A160 Peptide PCS-C18 column.
Conclusion
The BIOshell™ A160 Peptide PCS-C18 column delivers superior separation of basic compounds including peptides and proteins, as well as complex biologics like liraglutide, under weak ion-pairing conditions that are compatible with mass spectrometry. The BIOshell™ A160 Peptide PCS-C18 column facilitates rapid impurity characterization using both UV/VIS and MS detection platforms. The capability to achieve high-quality separation profiles with ion-pairing agents such as formic acid also minimizes environmental impact by removing the requirement for stronger fluorinated ion- pairing agents like TFA.
Related Products
HPLC Column
Solvents and Reagents
References
To continue reading please sign in or create an account.
Don't Have An Account?