Protein Expression Systems
Introduction
The development of genetic engineering and cloning has opened many possibilities of expression and isolation of heterologous proteins for research purposes. Considerable advances in technology have enabled expression and isolation of recombinant proteins in large scale. However, for large scale applications such as enzyme, antibody or vaccine production, the amount of protein required is considerably high. In such cases the system in which the protein is expressed must be easy to culture and maintain, grow rapidly, and produce large amounts of protein. Moreover, mammalian proteins also undergo various post-translational modifications. These requirements led to the discovery of protein expression systems. The various protein expression systems are bacteria, yeast, insect or mammalian systems.
The following factors determine the type of expression system used to produce recombinant proteins:
- time spent in expressing the protein
- ease of handling the expression system
- amount of protein needed
- mass of the protein
- type of post-translational modifications, number of disulfide bonds
- destination of the expressed protein
The process of expressing a recombinant protein in an expression system requires the following information/components.
- identification of the gene that encodes the protein of interest
- generation of cDNA from the respective mRNA
- selection of suitable expression vector to insert the gene sequence
- selection of suitable system that can express the vector
- appropriate screening and scaling up methods
The core steps involved in producing the desired recombinant protein are similar across the various expression systems (Figure 1).
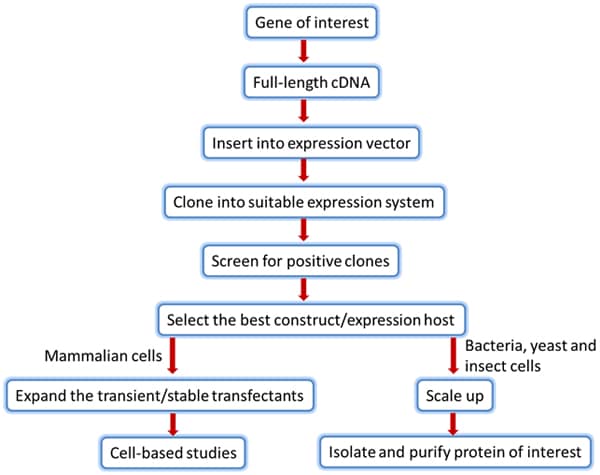
Figure 1.Steps involved in optimizing protein expression system to produce desired recombinant protein
Bacterial protein expression systems – Escherichia coli
Bacteria act as rapid and simple systems of expressing recombinant proteins due to the short doubling time. The media required to culture them are not expensive and the methods adapted to scale-up bioproduction are straightforward. The most widely used host system is E. coli since there is ample knowledge about its genetics, genome sequence and physiology. The genetic manipulation is easy and it also grows to high densities and is suitable for large-scale fermentations. However, the cell wall of E. coli contains toxic pyrogens and the expressed proteins may have to be extensively tested before use.
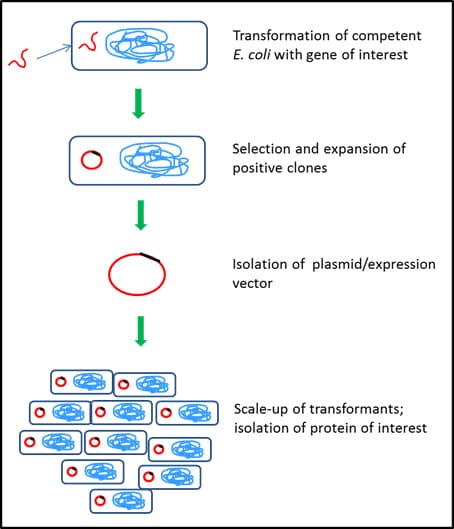
Figure 2.Bacterial protein expression system – Escherichia coli
Expression of protein using E. coli involves the following steps:
- use of competent E. coli (Catalog Numbers CMC0001, CMC0004, CMC0014) cells to take up DNA sequence of interest
- integration of the DNA into bacterial genome or circularization of the DNA sequence to exist as a plasmid
- selection of transformed E. coli using a selection marker (antibiotic) (Catalog Numbers L5667, L0168, L0543, L0418, L8795)
- expansion of selected E. coli to a higher scale in appropriate culture media, such as classic LB options (product numbers L2542, L3522, L3147) or EnPresso™ B Growth Systems (product number B11001)
- isolation and purification of intracellular/secreted proteins
Features
- low cost culture methods
- flexible system – can carry plasmids with multiple promoters, tags and restriction sites
- easy to scale up and produce higher yield of protein
Yeast protein expression systems – Saccharomyces cerevisiae
The highly developed genetic system, ease of use, reduced time input and costs have made S. cerevisiae an attractive organism for the expression and production of recombinant proteins. Yeasts are able to carry specifically designed plasmids and this ability is valuable in a recombinant protein expression system. The plasmid used consists of restriction sites that can be used to insert the gene sequence of interest. Transformation of yeasts with the plasmid produces the desired protein and can be appropriately scaled up.
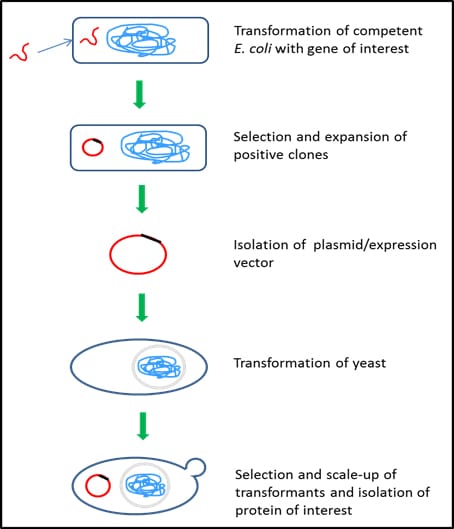
Figure 3.Yeast protein expression system – Saccharomyces cerevisiae
Expression of protein using S. cerevisiae involves the following steps:
- use of competent E. coli cells to take up DNA sequence of interest (Catalog Numbers CMC0001, CMC0004, CMC0014)
- integration of the DNA into bacterial genome or circularization of the DNA sequence to exist as a plasmid
- selection of transformed E. coli using a selection marker (antibiotic) (Catalog Numbers L5667, L0168, L0543, L0418, L8795)
- expansion of selected E. coli in appropriate culture media, such as classic LB options (product numbers L2542, L3522, L3147) or EnPresso™ Y Defined Media (product number Y22001)
- isolation of DNA or plasmid
- transformation into yeast (yeast transformation kit, Catalog Number YEAST1)
- screen the transformants for integration of DNA into yeast chromosome
- selection and scaling-up of high expressing yeast clones in appropriate culture media (Catalog Numbers Y1375, Y1501, Y1751, Y2001, Y1376, Y0750)
- isolation and purification of intracellular/secreted proteins
Features
- low cost culture methods
- suitable for both intracellular and secreted proteins
- provides eukaryotic post-translational glycosylation of proteins although it results in high
Insect cell expression systems – Sf9 and Sf21
The cell lines derived from Spodoptera frugiperda, Sf9 and Sf21, are frequently used as recombinant protein expression system. Baculovirus is a lytic, dsDNA virus, routinely amplified in cells of the insects belonging to Lepidoptera family. It is noninfectious in vertebrates and its promoters are inactive in mammalian cells.
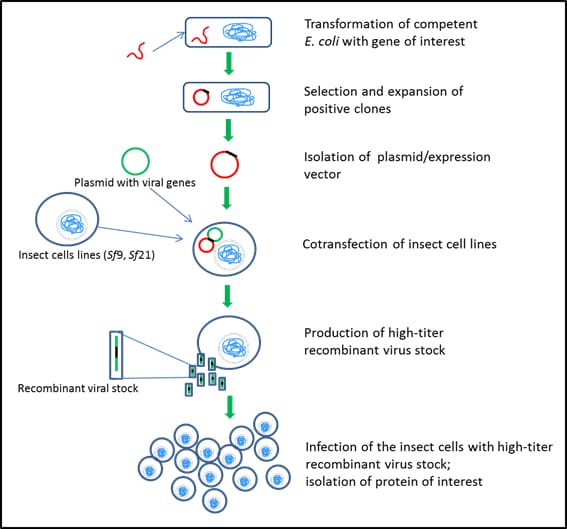
Figure 4.Insect cell expression systems – Sf9 and Sf21
Expression of protein using baculovirus/insect cells involves the following steps:
- use of competent E. coli cells to take up DNA sequence of interest (Catalog Numbers CMC0001, CMC0004, CMC0014)
- integration of the DNA into bacterial genome or circularization of the DNA sequence to exist as a plasmid
- selection of transformed E. coli using a selection marker (antibiotic) (Catalog Numbers L5667, L0168, L0543, L0418, L8795)
- expansion of selected E. coli in appropriate culture media (Catalog Numbers L2542, L3522, L3147)
- isolation of DNA or plasmid
- preparation of a second plasmid containing viral genes required for multiplication and formation of virus particles
- co-transfection of the expression plasmid and the second plasmid into Sf9 or Sf21 insect cells
- purification of the recombinant viral stock
- amplification of the virus and additional plaque assays to increase the titer of the recombinant viral stock
- infection of the insect cells with high-titer recombinant virus stock
- isolation and purification of intracellular/secreted proteins
Features
- recombinant protein is highly expressed during the last phases of lytic cycle before cell lysis
- suitable to generate both cytoplasmic and secreted proteins
- disulfide bonds in proteins are efficiently generated
- provide majority of post-translational modifications found in mammalian cells
Mammalian cell expression systems – HEK293 and CHO
The main challenge of using mammalian cells for expressing recombinant proteins is the reduced efficiency and levels of the protein expressed. However, cell lines such as HEK293 and CHO have been developed as efficient transient and stable expression systems, respectively. HEK293 cells are transiently transfected using liposomes, calcium phosphate or PEG as transfection reagents. Though transient expression is relatively easy and simple, scaling up is technically challenging. CHO cells are commonly used to stably express large quantities of recombinant proteins. The process involves transfection of DHFR-deficient CHO cells with the gene of interest and a DHFR selection cassette. The transfected cells are then screened in the presence of methotrexate to obtain stably transfected cell pools. The selection and expansion process takes 2-3 months
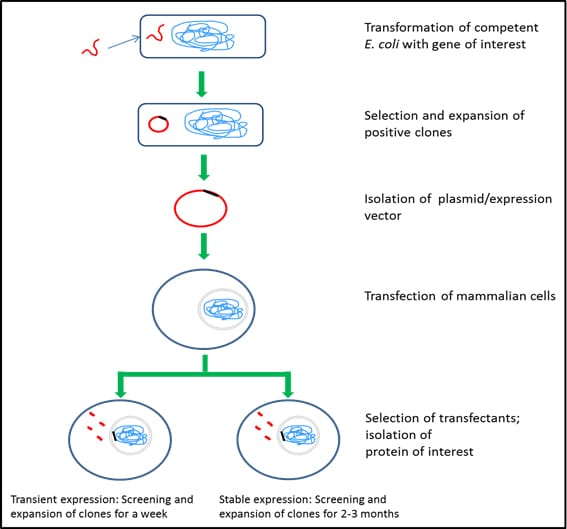
Figure 5.Mammalian cell expression systems – HEK293 and CHO
Transient or stable protein expression using mammalian cells involves the following steps:
- use of competent E. coli cells to take up DNA sequence of interest (Catalog Numbers CMC0001, CMC0004, CMC0014)
- integration of the DNA into bacterial genome or circularization of the DNA sequence to exist as a plasmid
- selection of transformed E. coli using a selection marker (antibiotic) (Catalog Numbers L5667, L0168, L0543, L0418, L8795)
- optional storage of clones in CloneStable™
- expansion of selected E. coli in appropriate culture media (Catalog Numbers L2542, L3522, L3147)
- isolation of DNA or plasmid
- transfection of the expression plasmid to mammalian cells using X-tremeGENE™ Transfection Reagents.
- selection of stable clones
- expansion of clones for transient batch expression OR expansion of clones for 2-3 months for stable expression
- isolation and purification of intracellular/secreted proteins
Features
- transient expression is easy and rapid
- provide all the post-translational modifications found in mammalian cells
- stable transfection results in higher yield, scalability and reproducible production
The following table compares the four protein expression systems and their respective advantages and disadvantages:
Choose an expression system that expresses the protein with all the desirable post-translational modifications. Irrespective of the choice, we offer a wide range of competent cells, insect and mammalian cell lines and related products that suit any protein expression system.
Materials
To continue reading please sign in or create an account.
Don't Have An Account?