Bis Tris Polyacrylamide Gel Electrophoresis Technology
Bis-Tris gel technology offers numerous advantages compared to traditional acrylamide gels for SDS-PAGE applications, including faster run times and sharper band resolution.
Polyacrylamide Gel Chemistry
PAGE uses a discontinuous buffer system, wherein the gel buffer ion differs from the running buffer ion. The difference in electrophoretic mobility between these two ions forms a moving voltage gradient that proteins travel through. Tris-Glycine gel chemistry is the most used PAGE system, which uses gels composed of Tris-HCl and running buffer composed of tris base and glycine. Tris-Glycine gels operate in a highly alkaline environment which can lead to undesirable protein modifications, such as deamination and alkylation. As a result, protein bands may be distorted or lose resolution in Tris-Glycine gels.
Bis-Tris Gel Technology
In contrast, Bis-Tris gels use Bis-Tris and HCl in the gel buffer and MOPS or MES in the running buffer. Bis-Tris gels operate at a neutral pH, minimizing protein modification and promotes protein stability during the gel run. This neutral pH leads to sharper protein band resolution and accuracy. Bis-Tris gels also have a longer shelf life than Tris-Glycine gels, which begin to hydrolyze over time. Bis-Tris gels have the flexibility to be combined with either MOPS- or MES-based running buffer; the difference in migration between these two ions results in different protein separation ranges. MES should be used when the protein of interest is small (<50 kDa), while MOPS should be used to resolve mid-to-large-sized proteins.
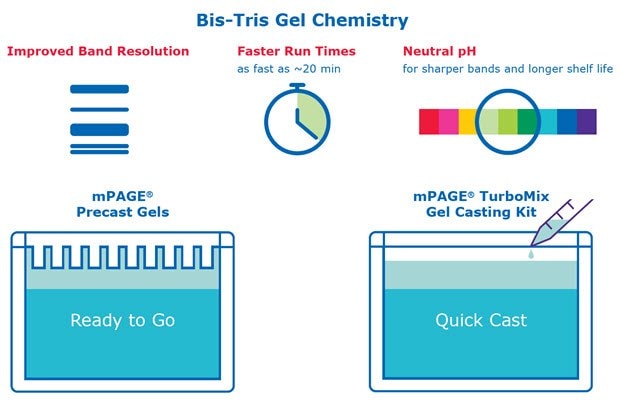
Figure 1.Bis-Tris technology features and benefits compared to traditional Tris-Glycine gels
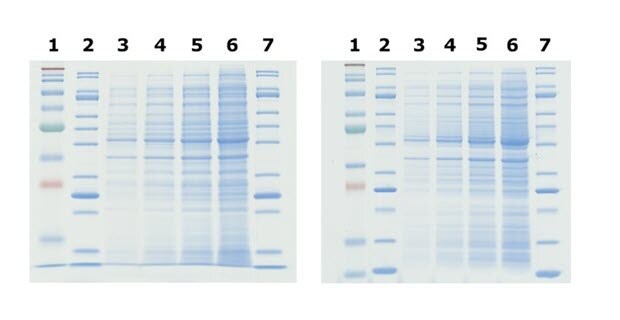
Figure 2.Comparison of Tris-Glycine (left) and Bis-Tris (right) gels. Tris-Glycine and Bis-Tris gels were hand-cast with 12% acrylamide and allowed to polymerize overnight. The gels were loaded with identical E. coli lysate titrations (lanes 3-6), mPAGE® unstained protein standard (lanes 2 and 7), and mPAGE® color protein standard (lane 1). The gels were run in either Tris-Glycine or MOPS running buffer, stained with ReadyBlue™ protein gel stain for one hour, and destained with deionized water for one hour.
Tris-Glycine and Bis-Tris Buffer Comparison
It is also critical to consider the type of sample buffer used during protein preparation. With Tris-Glycine gels, Laemmli buffer is typically used to denature and coat proteins in negatively charged SDS ions. Samples are then boiled at 100 °C to help facilitate denaturation. Heating Laemmli buffer to 100 °C causes the pH to become highly acidic. The combination of heat and acidity has been shown to cause protein cleavage, preferentially at Asp-Pro peptide bonds1. This cleavage leads to apparent protein degradation products during electrophoresis. Conversely, Bis-Tris gels use an LDS sample buffer that maintains an alkaline pH during sample preparation and does not require heating above 70 °C to fully denature proteins. This preparation maintains protein integrity by minimizing Asp-Pro peptide bond cleavage.
mPAGE® Bis-Tris Precast Gels for Protein Electrophoresis
The mPAGE® Bis-Tris SDS-PAGE Gel system offers high performance, optimal electrophoretic separation, and better resolution over a wide range of molecular weights. mPAGE® Bis-Tris Precast Gels have a versatile design that allows for larger sample loading volumes. The 10 x 8 cm mini cassette format makes mPAGE® Bis-Tris Precast Gels compatible with the most popular gel electrophoresis equipment.
mPAGE® Bis-Tris Precast Gels are designed to work exclusively with MOPS or MES running buffer. Depending on which running buffer is used, very distinct separation patterns can be achieved. MOPS buffer can be used to fine-tune the separation of large and medium-sized proteins, whereas MES buffer provides optimal separation of smaller proteins. Refer to the migration charts (Figures 3-7) to determine which gel running buffer system is best suited for the intended separation range.
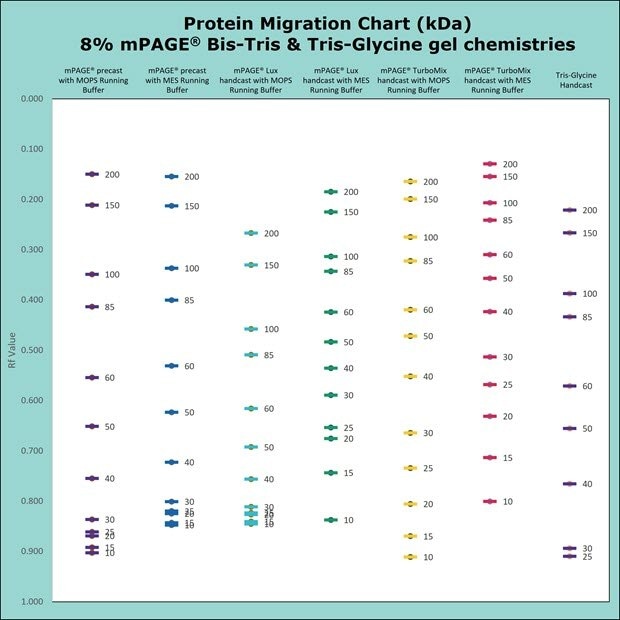
Figure 3.Migration Charts of 8% mPAGE® Bis-Tris & Tris-Glycine Gel Chemistries molecular weights displayed in kilodalton (kDa).
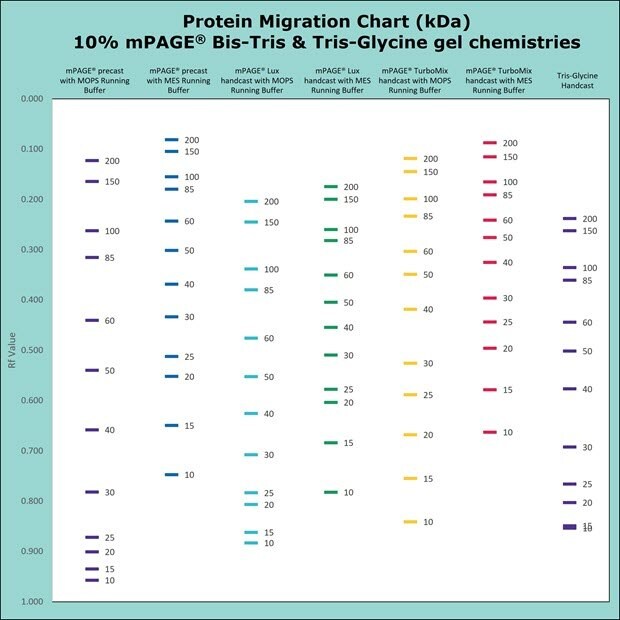
Figure 4.Migration Charts of 10% mPAGE® Bis-Tris & Tris-Glycine Gel Chemistries.
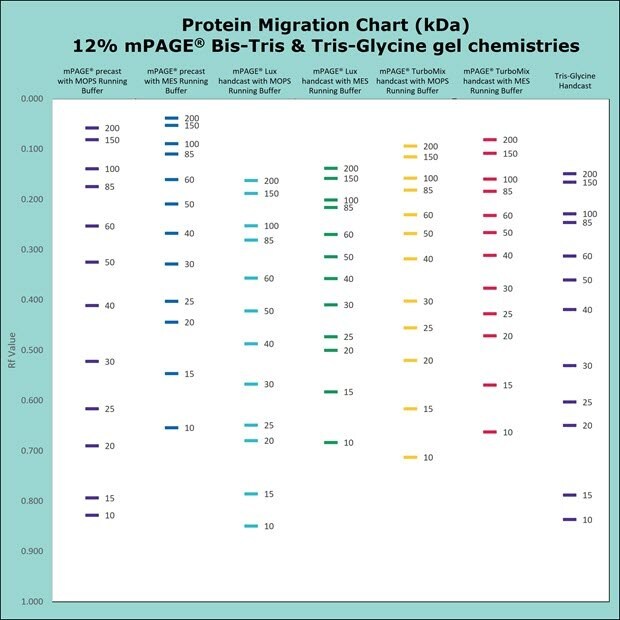
Figure 5.Migration Charts of 12% mPAGE® Bis-Tris & Tris-Glycine Gel Chemistries.
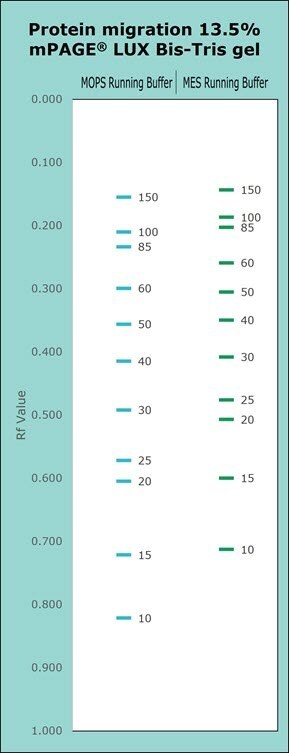
Figure 6.Migration Charts of a 13.5% mPAGE® Lux Bis-Tris gel with MOPS and MES running buffers.
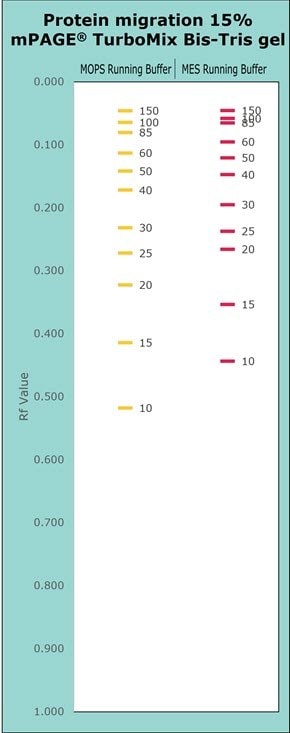
Figure 7.Migration Charts of a 15% mPAGE® TurboMix Bis-Tris Gel with MOPS and MES running buffers.
mPAGE® TurboMix Bis-Tris Gel Casting Kit
The mPAGE® TurboMix Bis-Tris Gel Casting Kit consists of a Resolving Solution and a Stacking Solution. The solutions are optimized to simplify gel preparation steps and minimize reagent waste. The mPAGE® TurboMix solutions can be used in traditional casting methods or the Quick Cast procedure (Figure 8).
The Quick Cast procedure polymerizes in one step by pouring the stacking gel immediately after the resolving gel. mPAGE® TurboMix Resolving Solution is provided at a 20% acrylamide percentage and formulated for dilution with deionized water, which allows the flexibility to cast different resolving gel percentages. The mPAGE® TurboMix Stacking Solution is provided at a 4% acrylamide percentage and requires only the addition of Ammonium persulfate (APS) and N,N,N’,N’-tetramethylethane-1,2-diamine (TEMED). For additional protein gel electrophoresis resources, please visit our Gel Electrophoresis application page for all of your protein resolution reagent and protocol needs.
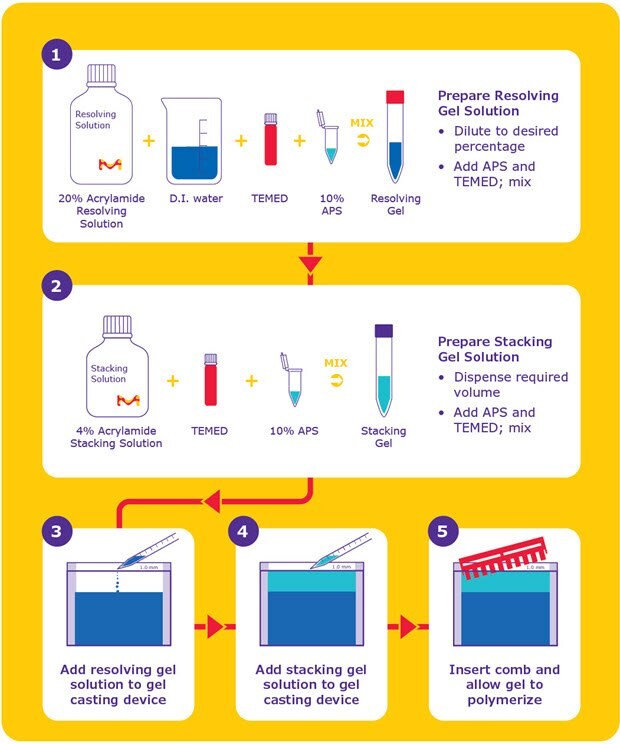
Figure 8.The mPAGE® TurboMix Quick Cast method offers a rapid, 5-step process for casting polyacrylamide gels.
Related Products
Reference
To continue reading please sign in or create an account.
Don't Have An Account?