Ephedrine/Pseudoephedrine HCl in Oral Solution Analysis
Ephedrine hydrochloride and pseudoephedrine hydrochloride can be used to reduce cold, allergic rhinitis, rhinitis and sinusitis caused by nasal congestion symptoms and control bronchial asthma. The same compounds are also found in the Chinese traditional medicine Xiao’er Kechuanling oral solution.
This application focuses on the determination of ephedrine HCl and pseudoephedrine HCl. Oral solution samples were distilled to extract the ephedrine HCl and pseudoephedrine HCl. Samples were then filtered through Millex® PTFE syringe filters, prior to HPLC-UV analysis using a Purospher® STAR RP-18 endcapped HPLC column.
The limit of detection (LOD) and the limit of quantitation (LOQ) were 0.04 µg/kg and 0.12 µg/kg, respectively, for ephedrine, and 0.02 µg/kg and 0.06 µg/kg, respectively, for pseudoephedrine. The method can be used for determination of ephedrine HCl and pseudoephedrine HCl in Xiao’er Kechuanling oral solution.
Figure 1.Chemical structures of compounds used in the study.
Materials
Determination of Ephedrine HCl and Pseudoephedrine HCl in Xiao’er Kechuanling Oral Solution
Purospher® STAR RP-18e

Figure 2.Chromatogram of ephedrine HCl and pseudoephedrine HCl standard solution.
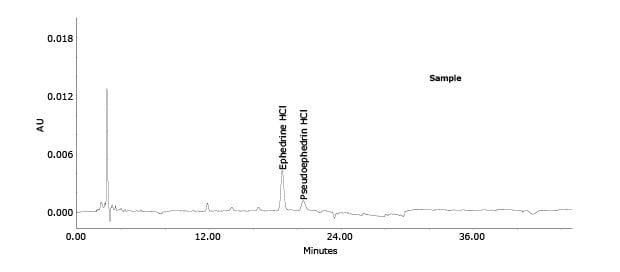
Figure 3.Chromatogram of in Xiao’er Kechuanling oral solution.

Figure 4.Chromatogram of solvent blank.
Specificity and Repeatability – Ephedrine and Pseudoephedrine
Calibration Data - Ephedrine

Figure 5.Calibration curve of ephedrine HCl.
Calibration Data - Pseudoephedrin

Figure 6.Calibration curve of pseudoephedrine HCl.
Conclusion
The method can be used for ephedrine HCl and pseudoephedrine HCl in Xiao’er Kechuanling oral solution.
如要继续阅读,请登录或创建帐户。
暂无帐户?